TECH-ENABLED
ACCELERATION.
Meditrial is a leading Clinical Research Organization (CRO) dedicated to Clinical Trials, Medical Education, Regulatory Affairs and Market Access.
Meditrial drives acceleration with leading edge technology and digital assets to reach milestones with simplicity, speed and efficiency.
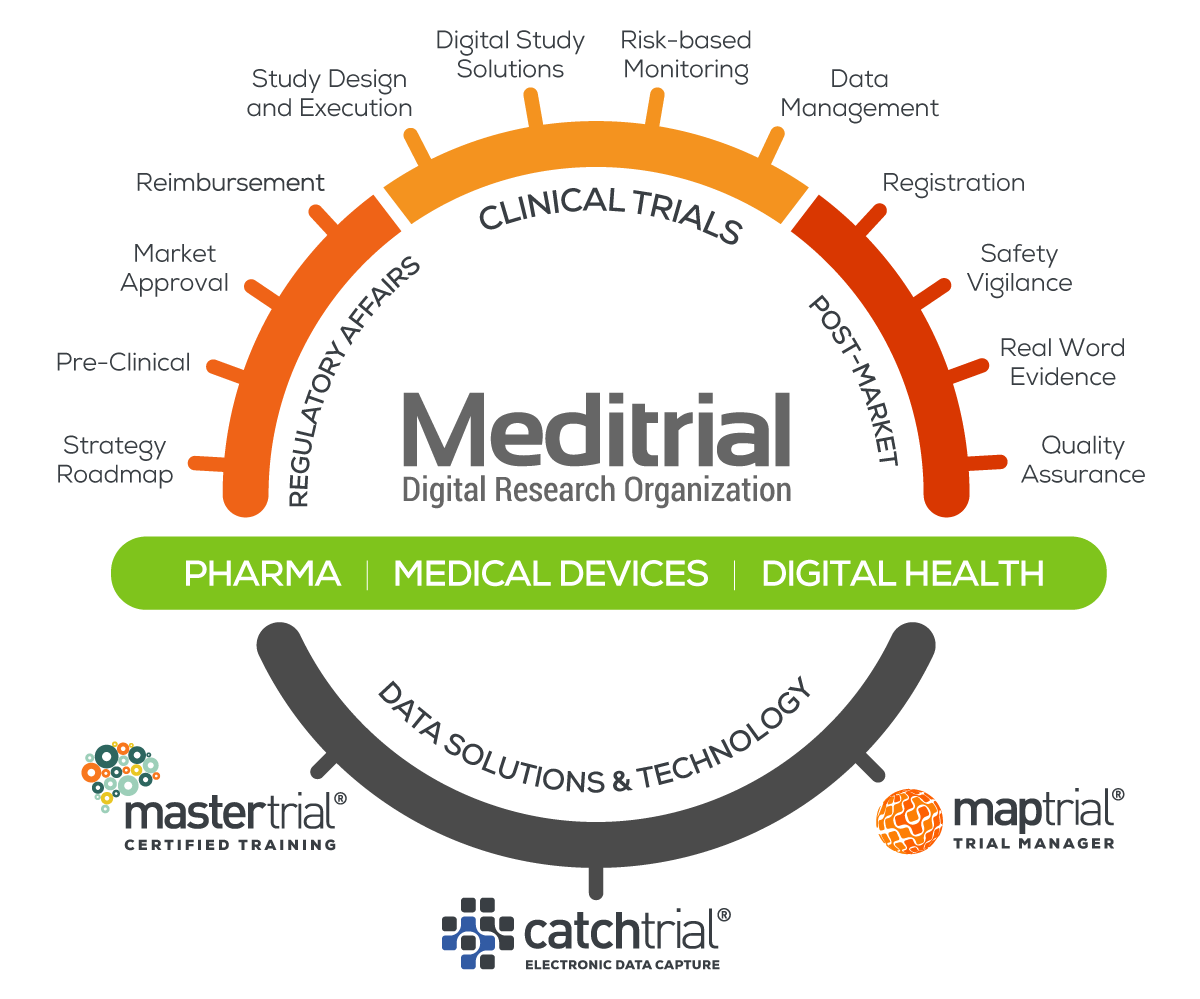
SOLUTIONS & TECHNOLOGY
PHARMA
Meditrial helps pharmaceutical clients develop future focused business strategies and to implement the time critical programmes and procedures essential to success within worldwide regulatory frameworks.
MEDICAL DEVICES
Meditrial is a leading, full services Clinical Research Organization (CRO) focused on the needs of the medical device industry. We are proud to serve the most innovative medtech companies in the world.
BIOTECH
Meditrial culture and operating structure are purposely designed to accommodate efficient partnering, important for emerging biotechs all while delivering necessary results to investors, stakeholders and regulatory authorities.











