CLINICAL SOFTWARE.
DIGITAL HEALTH SOLUTIONS


CATCHTRIAL
To manage and control your study, Meditrial’s innovative team brought you cloud-based complete data management system Catchtrial. All in one CRF, Data Validator, Report Builder, Core Lab images and Safety Adjudicator to build your global trials.
Catchtrial is ready to support worldwide medical research by capturing clinical trial data and images with instant, advance and on-the fly translation cloud-based platform for every stakeholder associated with your study.
CENTRALIZED BIOMETRICS
ELECTRONIC DATA CAPTURE
Today, immediate clinical data access and decision making are key to success. Meditrial combines exceptional expertise for data management with a proprietary software option for real life Electronic Data Capture (EDC).
Capture your trial data directly on an iPad with Catchtrial. With instant data validation, clean data is available in seconds allowing immediate decisions from anywhere in the world.
Through our proprietary system and technology platform, we optimize your data and processes, ensuring fast and efficient marketing of your products.
We understand the industry and how to make the most of advanced technologies.
Reach out for a consultation on how we can work with you to reduce your time to market, expand your marketability and reach a more meaningful value-based outcome through design and strategy.

CLINICAL TRIAL MANAGEMENT SYSTEM
By providing a single, centralized system to orchestrate operational and administrative activities, Maptrials CTMS allows sponsors and CROs to intelligently manage the complexities of clinical trials.
MAPTRIAL
THE ULTIMATE TOOL FOR TRIAL MANAGEMENT
Maptrial CTMS (Clinical Trials Management System) yields immediate efficiencies and cost savings for clinical operations. By providing a single, centralized system to orchestrate operational and administrative activities, Maptrial allows Sponsors and CROs to intelligently manage the complexities of clinical trials. Product modules offer comprehensive management of trial planning, country and site progress, monitoring activities, supplies and finance, with state-of-the-art investigator data mining and operational support.
For single-site and multi-site organizations, Maptrial is a cloud-hosted clinical trial management system (CTMS) designed to track the operational data of clinical trials across any therapeutic area.

MASTERTRIAL
THE NEW WAY TO MASTER EVOLVING REGULATIONS
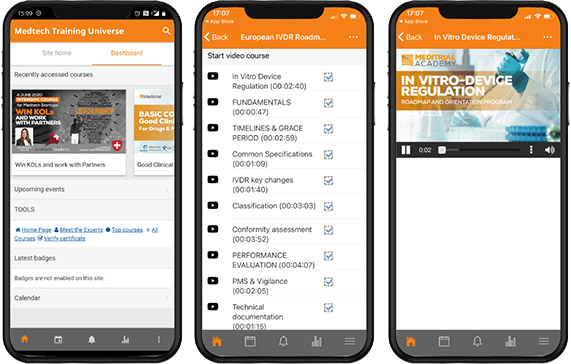
Meditrial Academy – Learn the latest topics in the medical devices arena.
Our Global Online Education, exclusively dedicated to Meditrial customers.
During your journey into the Meditrial World you will find the best medical devices e-learning solutions. Each course provides interactive toolbox, simple tutorials, video lessons and final quizzes to guide you step by step into the MedTech universe.












