MARKET ACCESS.
ACCESS KEY MARKETS
MARKET ACCESS SERVICES
Meditrial is the Representative trusted by more than 100 firms.
Market access has never been easier.
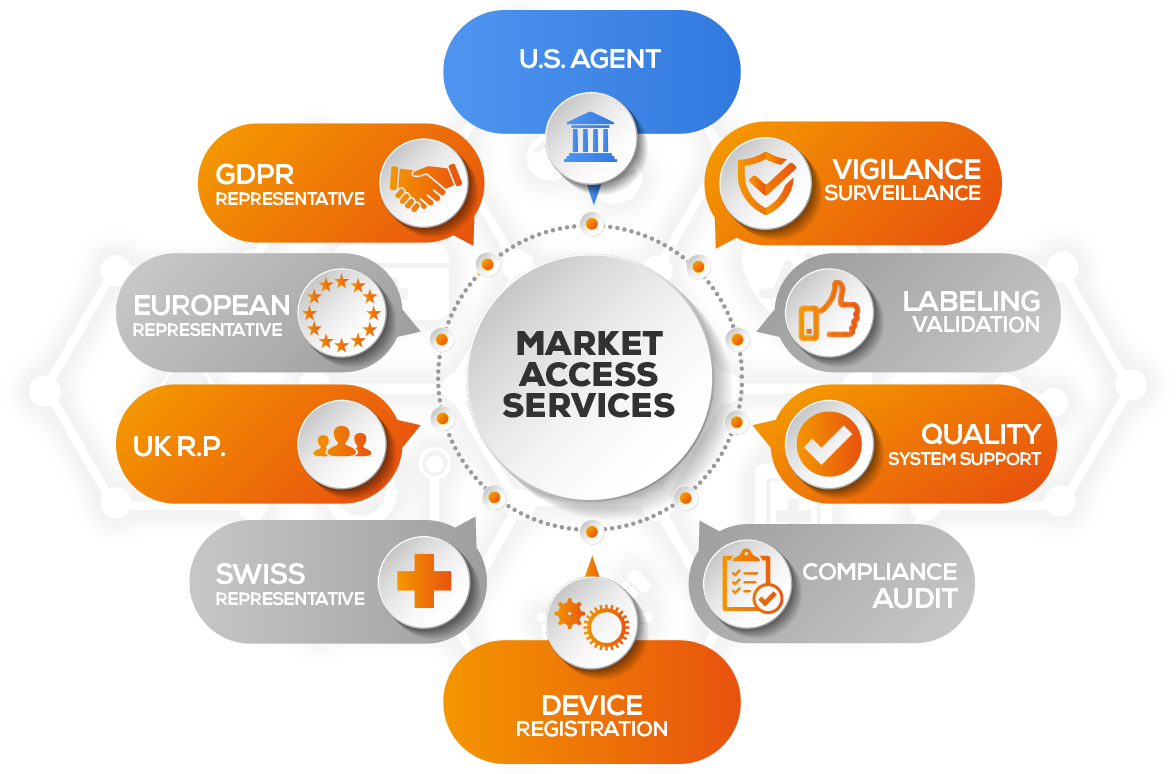
MARKET ACCESS
MARKET ACCESS & COMMERCIAL SOLUTIONS
Meditrial’s comprehensive suite of commercial services powered by Data & Analytics drive value creation and commercial success.
Create informed strategies from early development through commercialization
Improve patient outcomes and identify opportunities to lower total cost of care
Inform healthcare and clinical decision-making to accelerate access
Generate regulatory-grade evidence to quantify the economic and clinical value
- Market Landscape Assessment
- Early Commercial Evaluation
- Market Segmentation
- Stakeholder Mapping
- Payer, Provider and Patient Surveys
- Policy Research, Analysis and Strategy
- Coverage, Coding and Reimbursement
- Competitive Analysis, Pricing and Payment
- Health Economic and Financial Models
- Claims Analysis and Payment Modeling
- Research on HTAs and Clinical Trials
- Real World Evidence
- Observational Research & Surveys
- HEOR Strategy and Gap Analysis
- Manuscript Writing, White Paper & Green Book
- Global (multi-country) Access Strategy
- Coding and Reimbursement Applications















