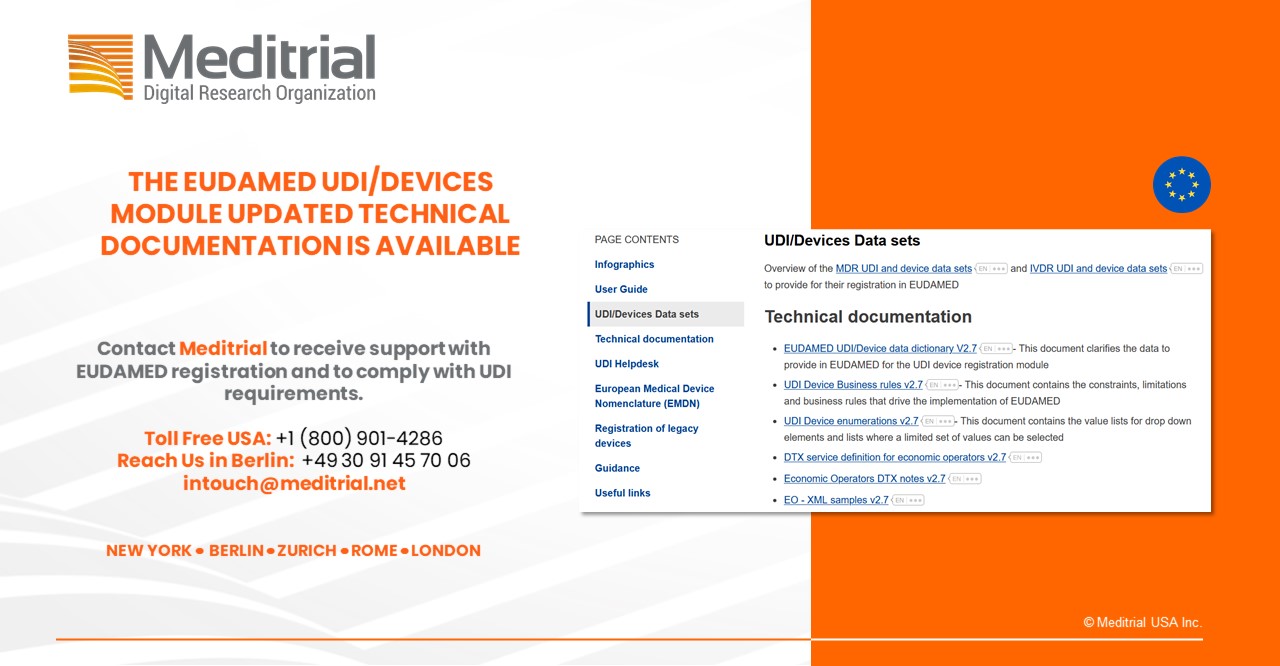
The European Commission published a series of new UDI device related documents.
- UDI/Devices User Guide for Economic Operators
- EUDAMED UDI/Device data dictionary V2.7
- UDI Device Business rules v2.7
- UDI Device enumerations v2.7
MDR and IVDR introduce an EU device identification system based on a unique device identifier (UDI) allowing easier traceability of medical devices. This requires that manufacturers submit in EUDAMED the UDI/Device information of all devices they place on the EU market. Manufacturers can already enter UDI/Device information in the system on a voluntary basis.
Relevant documents and links on UDI/Device registration published by the EC.
Do you know how to register for EUDAMED? Contact Meditrial to learn everything you need to know on how to prepare! With us, you will be the first to comply.
Contact us for immediate assistance in Europe or the US.







Leave A Comment