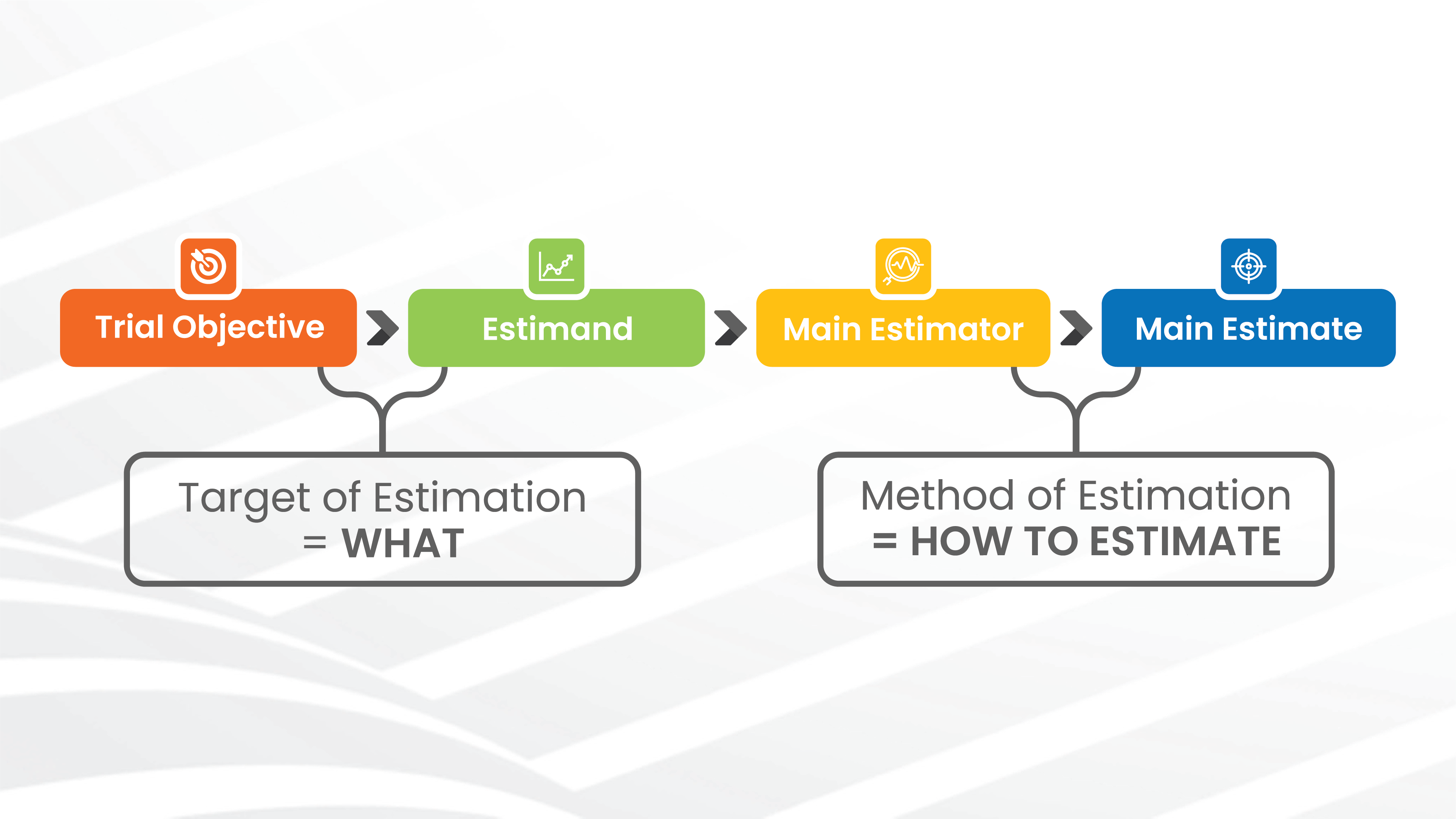
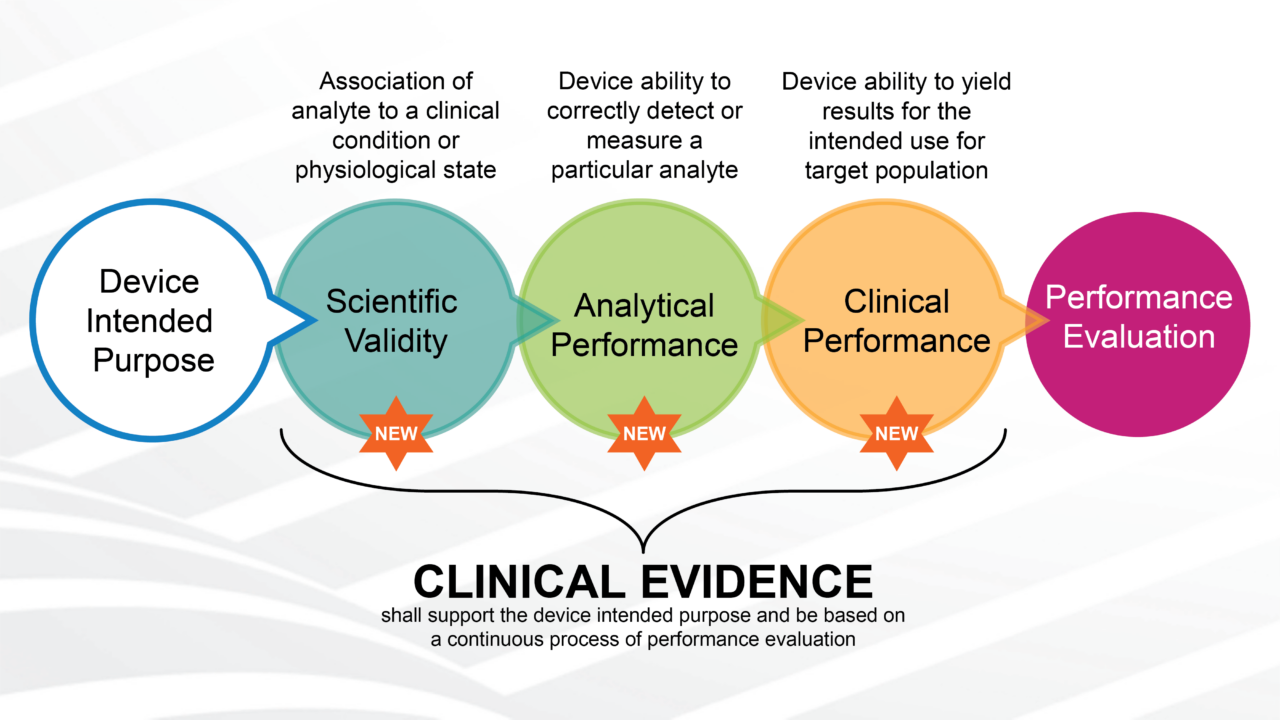
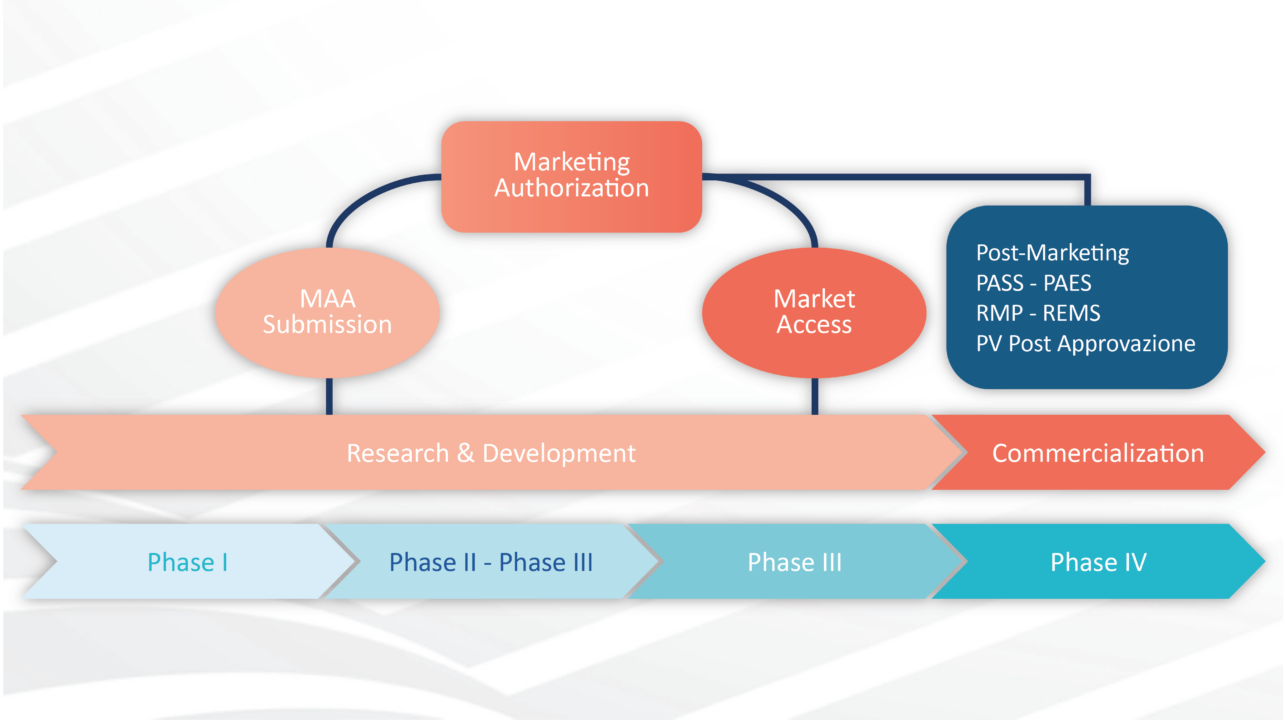
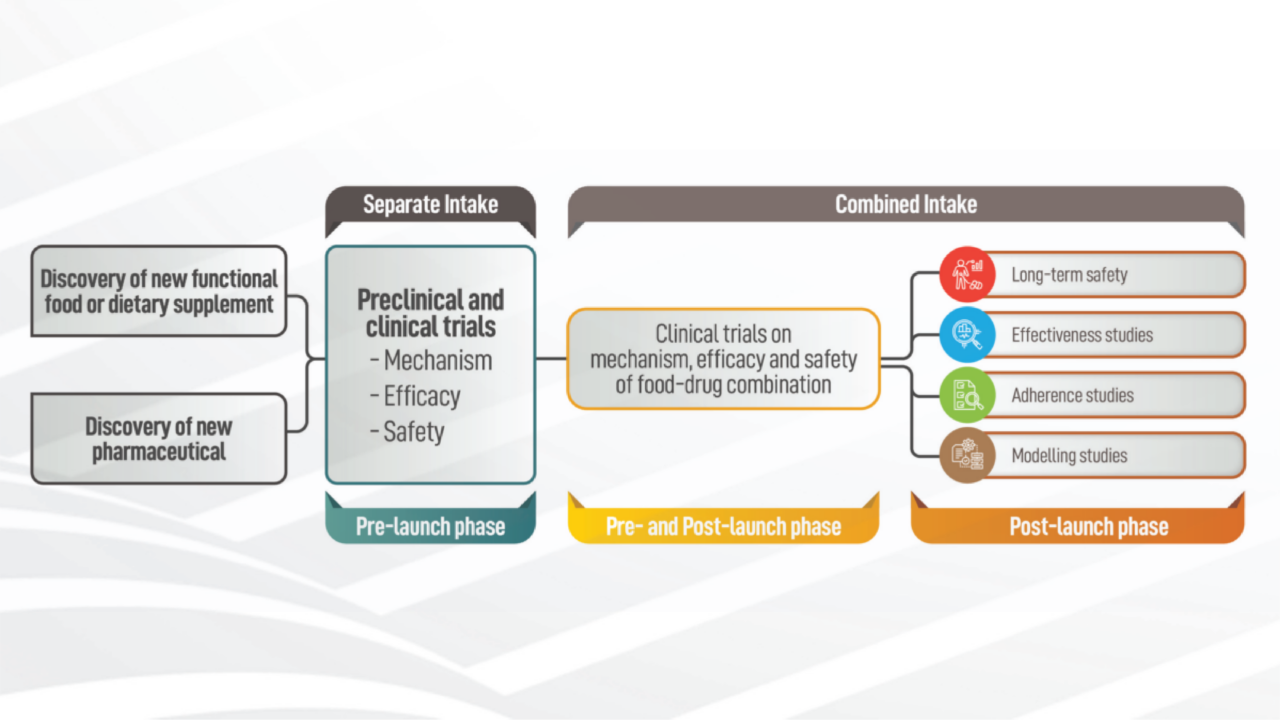
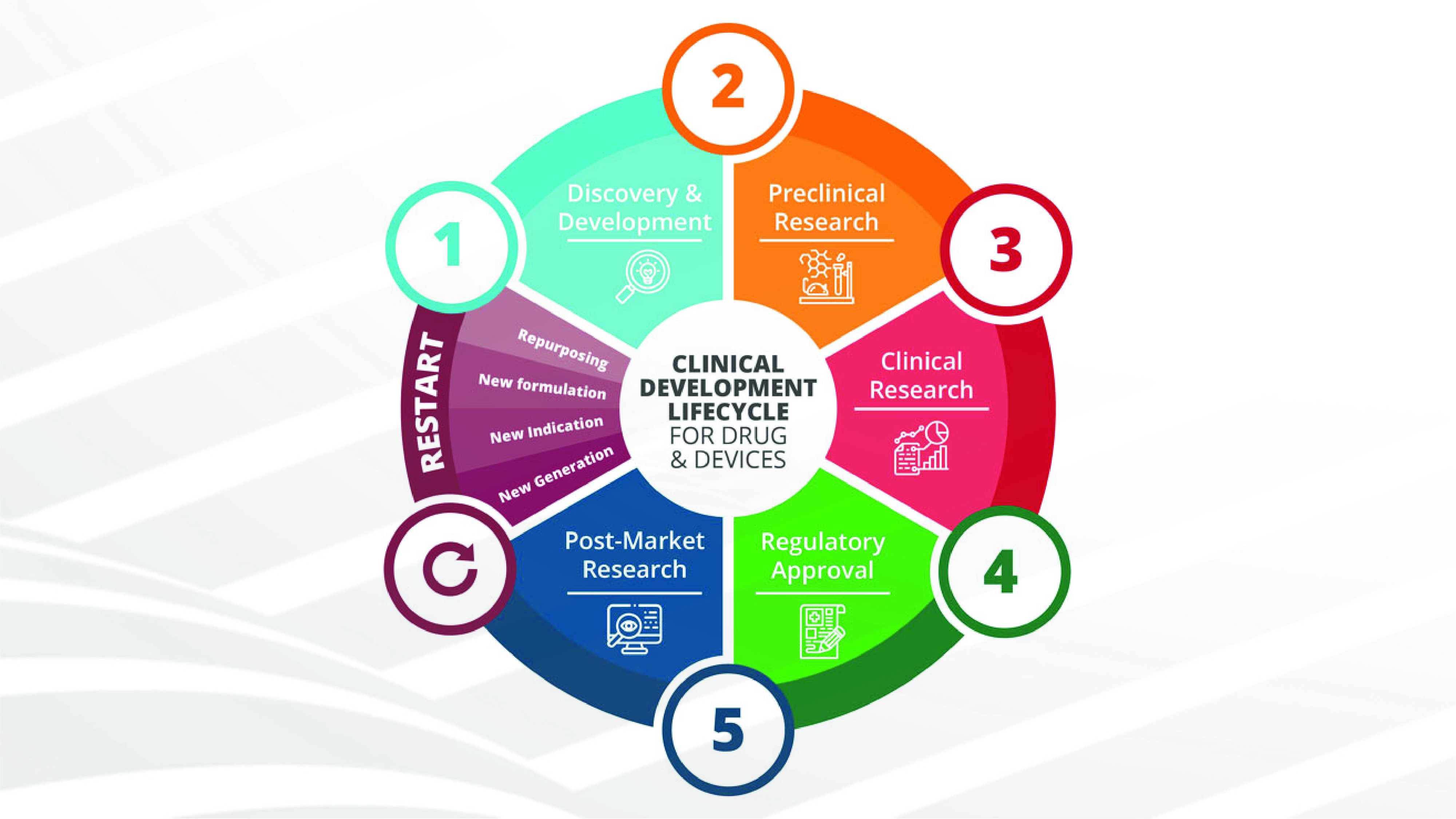
CLINICAL TRIAL DESIGN: THE REVOLUTION OF NEW STATISTICAL PRINCIPLES
Edited by Dr. Monica Tocchi, Meditrial medical director. The organization called ICH (International council for harmonization of technical requirements for pharmaceuticals for human use) is a global organization that unites regulatory authorities and the pharmaceutical industry in the common [...]