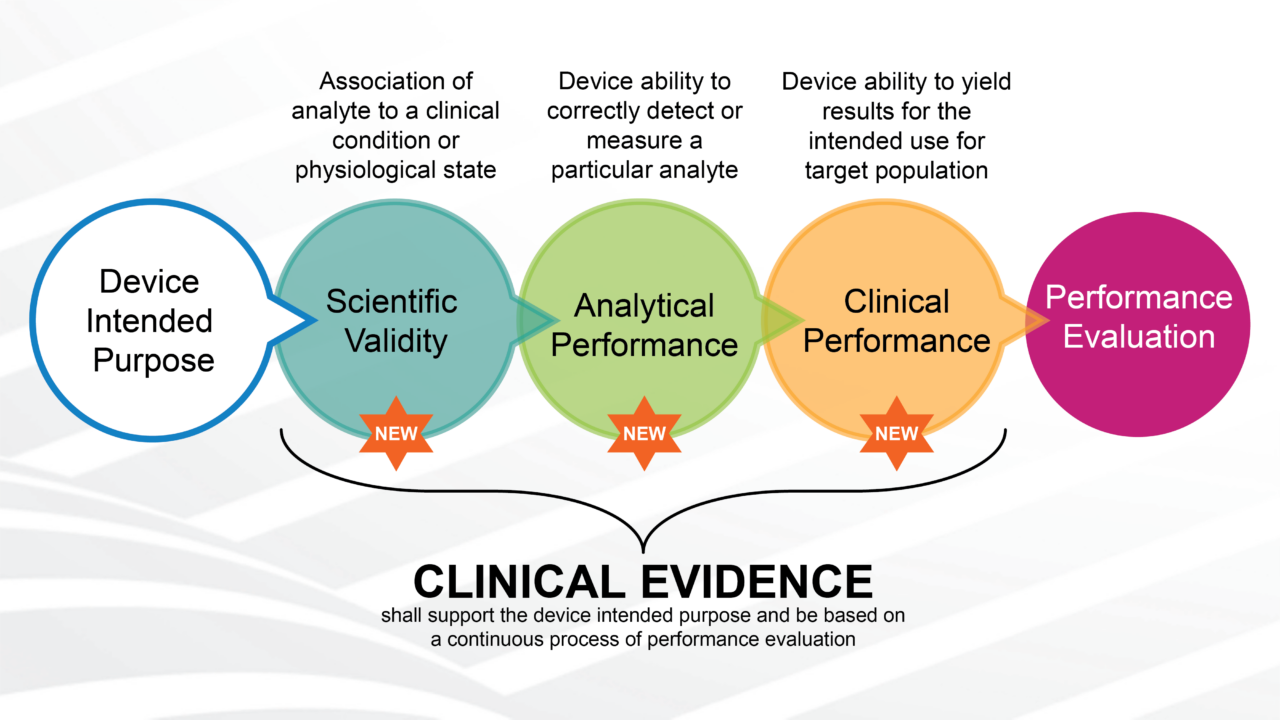
CLINICAL EVIDENCE FOR IVDs: THE NEW GOOD CLINICAL PRACTICES
Atty. Alessia Nuciari, director of compliance Meditrial. The in vitro diagnostics industry has undergone a substantial transformation. IVDR Regulation 746/2017, published in May 2017 and applied from May 26, 2022, introduced major changes in the industry. The main [...]