EUDAMED UDI/devices Updated Technical Documentation Module
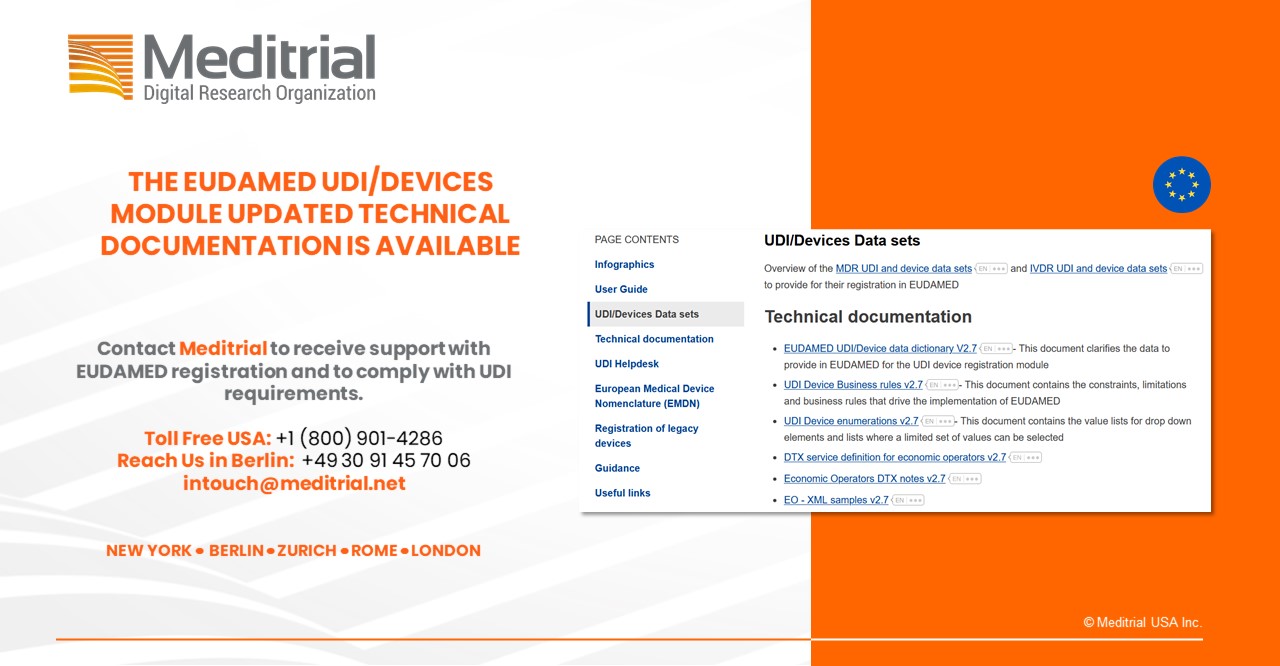
The European Commission published a series of new UDI device related documents. UDI/Devices User Guide for Economic Operators EUDAMED UDI/Device data dictionary V2.7 UDI Device Business rules v2.7 UDI Device enumerations v2.7 MDR and IVDR introduce an EU device [...]