Historically, patient organizations have had the primary purpose of supporting the communities they represented. Today, this remains a crucial function, but modes have changed, as has the role that associations and patients themselves take on in clinical research.
The WHO Alma-Ata Declaration (September/1978) emphasized that people have the right and duty to participate individually and collectively in the planning and implementation of their own health care. This means that people must be involved not only in the delivery of health care, but also in the way drugs are researched, developed and brought to market. This can only be achieved through a transparent and continuous dialogue between patients and industry. In this way, the patient can become a central figure, taking their demands all the way to the development and research stages.
In this sense, patient associations have taken an increasingly active role in clinical trials and have rapidly evolved in their specific areas of interest and the level of expertise of their members. Therefore, associations are increasingly recognized as partners in the world of clinical research. New trends are emerging in this area, such as the centrality of the patient and his or her primary role in influencing the health and ethics of research: stakeholders and from institutions believe that associations can therefore channel the voice of those they represent while strengthening their role in various research activities.
The active involvement of participants in clinical trials and thus of associations in the planning and execution of clinical trials is crucial, not only for the actual success of the study, but also for the communities and study participants to benefit effectively.
A Winning Combination
In clinical trials a crucial aspect is the initial design, which can have a major impact on the final outcome. Regardless of the efficacy of the treatment or device, it is the study as a whole that must be designed to be acceptable to patients. To this end, associations take patient instances all the way to the early stages of study planning and design, dialoguing with pharmaceutical companies to ensure that their needs are indeed well understood and placed at the center of their study.
However, the involvement of patient associations in clinical trials often occurs too late and is sometimes overlooked altogether. This leads to clinical trials focused solely on solving clinically relevant challenges and achieving endpoints, without assessing the feasibility of the trial itself in light of the difficulties involved for the participating subject.
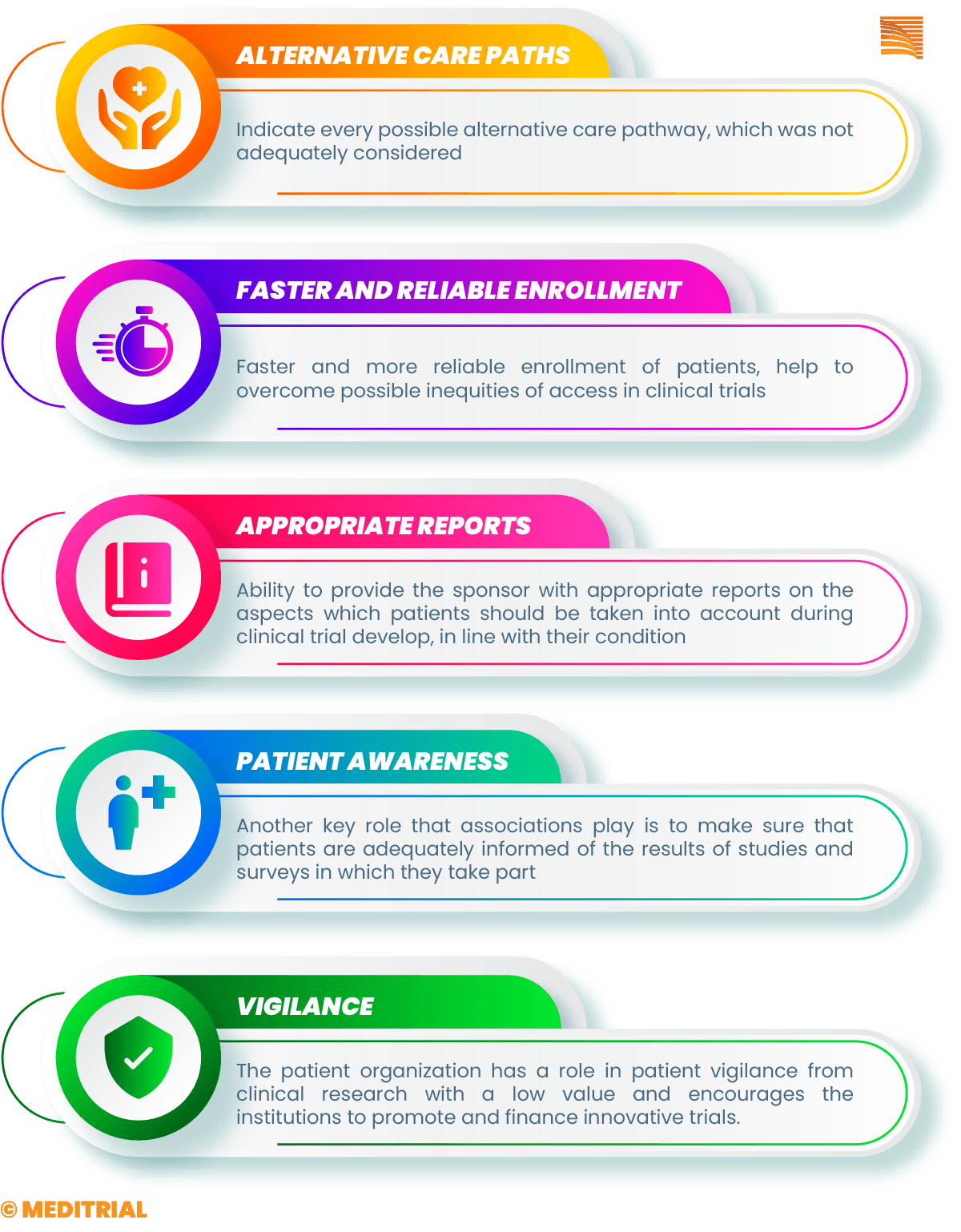
Conversely, greater attention should be paid to addressing the priorities, preferences, and needs of patients and their quality of life, which is already compromised due to disease symptoms or treatment burden. Patient associations can be crucial in providing a true measure of the impact of the drug or device study on the patient’s quality of life and possibly suggest alternative pathways in the study design not adequately considered.

Involvement must be Early
Il coinvolgimento delle associazioni nelle prime fasi dello studio può inoltre rendere l’arruolamento più rapido e affidabile, nonché aiutare a superare possibili iniquità di accesso nei trial clinici, che costituiscono per molte patologie l’unica possibilità per migliorare la propria condizione patologica. Le associazioni dei pazienti hanno la capacità di favorire il reclutamento dei pazienti nello studio grazie a una conoscenza ampia e approfondita della dislocazione dei pazienti sul territorio e delle loro condizioni.
The importance of PROs
Associations also play a key role after patients have been enrolled and when the study has already started, as through constant and direct support from patients, they can better capture difficult conditions, curb them, and thus reduce “drop-out rates.” To this end, the associations advocate the inclusion of patient-reported outcomes (PROs) in the study endpoints. PROs can allows the focus of the study to be shifted more toward the patient as the major stakeholder in the study outcomes.
In addition, patient associations present themselves to sponsors as a point of reference in providing reliable and timely information about the communities they represent and are also able to provide pharmaceutical companies with reports that are useful in identifying aspects of the study that can best meet the primary needs of patients.
Dissemination of information and supervision of trial results
Another key role that associations play is to make sure that patients are adequately informed of the results of studies and investigations in which they take part. Associations can disseminate study results in ways that are easy to understand, hold meetings with patients to share the results, and make sure people understand the implications.
Of course, associations play a watchdog role in protecting patients from lower-value clinical research, or conversely, they can push institutions to promote and fund particularly innovative trials. In addition, more and more representatives of patient organizations are included in official bodies, advising on health policy and care decisions.


Best Practices of partnerships in clinical trials
A review of the literature shows that there is little empirical data to define and optimize the relationship between patient associations and sponsors in clinical research, but some general principles that are critically important for positive collaboration between the parties appear to be well understood. These principles can be summarized as follows:
- Identify common goals. Pharmaceutical and medical device companies and patient organizations should be clear about the purpose of the study and the common goals to be achieved;
- Transparency. Relationships between associations and pharmaceutical companies should be totally transparent, including the financial aspects of the study;
- Independence of associations in all their decision-making as a guarantee of credibility and trust for patients;
- Adequate understanding of the benefits that all study stakeholders and especially patients can gain from fruitful collaboration for the purpose of clinical research.

At the same time, to strengthen their action in the field of clinical research, patient associations must overcome several obstacles such as: exclusion from discussions with regulatory bodies, the risk of undue or inappropriate influence by companies on their decision-making process, and the risk that collaboration with pharmaceutical companies may be perceived by the community as motivated by commercial interests.
Patient associations to overcome these obstacles and for appropriate involvement in clinical trials can adopt some useful and effective best practices:
- Support fundraising for scientific studies. Some associations cooperate with scientific societies in raising funds, which in turn scientific societies can direct into research.
- Collaboration with pharmaceutical companies is a crucial aspect, and this can be facilitated by increasing the strength of representation by merging and/or federating multiple associations, such as the shining example of the Global Heart Hub, by making specific transparency commitments useful to avoid potential conflicts of interest.Synergy with policy and institutions through advocacy activities aimed at influencing decision-making and legislation and thus resource allocation toward specific scientific studies.
- Synergy with policy and institutions through advocacy activities aimed at influencing decision-making and legislation and thus resource allocation toward specific scientific studies.
- The use of appropriate digital communication channels can facilitate an ongoing and more effective dialogue with sponsors.

As this brief analysis shows, many obstacles remain to be addressed in the integration and collaboration between patient associations and clinical trial sponsors.
However, it emerges equally clearly how the full and genuine collaboration of sponsors and even academia with associations can only make a favorable contribution to the success of clinical trials.







Leave A Comment