Clinical development is a term traditionally used to define the entire process of bringing a new drug or medical device to market. It includes a pre-clinical phase to support human use and a clinical trial phase on patients, in compliance with current regulations. But today, after 29 months marked by a deadly pandemic, the global healthcare ecosystem has been completely and irreversibly transformed. New technologies have revolutionized vaccine development, overcoming the limitations of traditional approaches (Cell). The response of governments around the world to the pandemic, with regulatory authority initiatives to accelerate evaluation procedures for Covid-19 diagnostic tests, treatments and vaccines, has unlocked unprecedented opportunities in clinical development. At the same time, the wave of technological progress in response to the pandemic has also accelerated a convergence in drug and technology development processes (Figure 1).
THE NEW ROADMAPS
For the clinical development of drugs and medical devices
For the clinical development of drugs and medical devices
We are approaching the point where many countries are likely to declare some sort of victory over Covid by classifying the disease as ‘endemic’, but the risk is still very high (Nature). Various elements could reverse all the progress of recent months and cause Covid cases and deaths to rise again, potentially prolonging the pandemic into its third year and beyond. It is therefore necessary to confront new and dynamic changes in health services, demand and capacity, which require different solutions from those adopted in the past.
“This column aims to highlight emerging trends and new roadmaps for clinical development,” comments Dr. Monica Tocchi, medical director and founder of CRO Meditrial. “With a pragmatic approach and in collaboration with Italian and international experts, we will discuss multiple topics with the aim of providing elements of interest not only to clinicians and practitioners, but also to patient associations, governmental authorities, scientific societies, non-profit organizations and all other actors involved in the health system value chain”.
CONTINUOUS EVOLUTION
Changing health needs and societal expectations, emerging innovations, and changes in the regulatory environment will transform the clinical development process in the near future.
As the development process of both new molecules and medical devices or diagnostics is highly articulated, complex and costly, it is appropriate to consider the strategies available to foster efficacy, best use of resources and return on investment. In this context, clinical development can be considered as a continuous cycle; after the typical five phases (shown in Figure 1), the development process can be restarted and completed more quickly, due to the potential to reposition existing drugs or technologies for new therapeutic purposes, or to introduce new formulations, expand use with new indications or create new generations of the product, an approach typical of medical devices that are regularly subject to technological improvements.
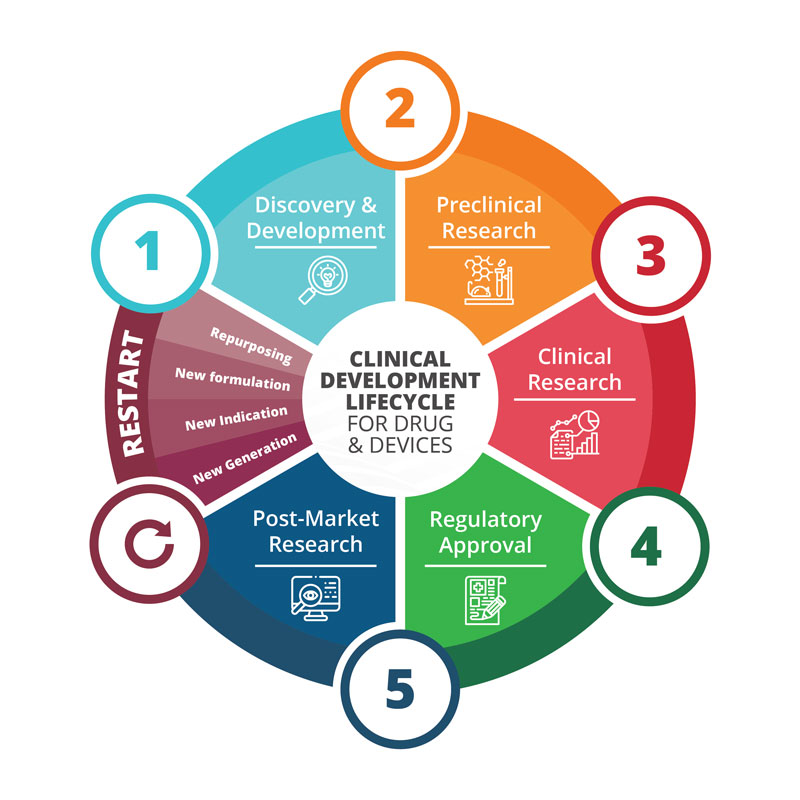
Figure 1.
Convergence of clinical development for pharmaceuticals and medical devices. The lifecycle starts with research and development (1), continues with preclinical research (2), extends with clinical studies in humans (3), to achieve regulatory approval (4) and expands with the launch and marketing phase (5). The life cycle starts again in the case of repurposing, or the introduction of new formulations, new indications or new generations of the product.
NEW APPROACH TO CLINICAL TRIALS
Digital technology for conducting the clinical trials necessary to create the evidence of benefit and safety, the complexity of designing protocols globally, the need to structure decentralized studies by putting the patient at the center of the study and making him or her an active participant in the development process, are among the factors that have the greatest impact on the development pathway of drugs and devices, as well as other types of products such as biotechnology or nutraceuticals.
Quoting a recent report by the National Institute of Health, ‘partnership’ is the key word for making clinical research easier for patients and faster (ISTISAN Reports 22/4 EN). In today’s global and interconnected world, innovation, development and well-being are achieved by working ‘together’.
“The success of clinical trials depends not only on the doctor-patient connection, but on countless other relationships and joint efforts by manufacturers, sponsors, ethics committees, competent authorities, centers, foundations, researchers, legal offices, and participants, who cooperate to synergistically enable medical progress in the presence of increasingly stringent international standards and guidelines,” Dr. Monica Tocchi further emphasizes. “Our daily activity as research professionals is aimed at creating a more effective collaboration between the medical community, patients, the healthcare industry and regulatory authorities.”
THE NEW GLOBAL ECOSYSTEM
The pandemic has presented an exceptional opportunity to challenge current scenarios and the way we conduct clinical research, bringing about a radical change in all traditional processes, encouraging active collaboration of all parties involved. This new column will address the challenges and opportunities in a practical and clear way, outlining possible solutions affecting the future of research and science.
At Meditrial, we firmly believe that the profound change in the healthcare ecosystem urgently requires the need to share our collective knowledge to help the sector achieve new goals resulting from the fast evolving rules and regulations, the unstoppable global computerization and the irreversible transformation of our society’s needs and requirements.
In the Rubric we will make use of illustrative graphics, multimedia and digital audio-video tools to expose the concepts covered with immediacy. We will provide a blog and social media to stimulate discussions and create a dialogue aimed at adapting and adopting the new paradigms of today and tomorrow.







Leave A Comment