Italy has recently instituted substantial changes in national regulations on medical device vigilance in order to align our procedures with European Regulations 2017/475 for medical devices and 2017/476 for in vitro diagnostics. As of Oct. 16, 2022, the new rules of the March 21, 2022 Decree “Establishment of the national device-vigilance network and the information system to support it” published in the Official Gazette on April 16, 2022, will be mandatory.
Public and private healthcare professionals and manufacturers, and even “lay users” of medical and diagnostic devices, will be required to make reports of incidents, malfunctions and medical errors to the Ministry of Health, for the first time through a new electronic system. In fact, a dedicated IT system is planned to support the reporting network within the New Health Information System (NSIS) portal. In this article we look at the main changes and how to prepare for the adoption of the new rules, with health legal considerations from an expert, Prof. Guerino Fares, head for Crispel University Roma Tre of the “Pharmacy and Medical Devices” area.

THE RISKS ASSOCIATED WITH MEDICAL DEVICES
Medical devices are rapidly increasing in variety and complexity and are a mainstay of patient care because in many cases they are essential aids to survival or improve health and quality of life. Medical devices encompass a wide range of health or medical tools essential for the prevention, diagnosis, cure, or treatment of an abnormal disease or physical condition. Worldwide regulations identify different categories of devices, based on the level of risk involved in using the device. Those considered highest risk, are those associated with a greater likelihood of causing serious or adverse health consequences. Examples of high-risk devices include lung ventilators, pacemakers, and artificial heart valves. Among in vitro diagnostics, high-risk devices are those that detect or are exposed to life-threatening transmissible agents and infectious diseases with a high risk of propagation.
Before being placed on the market, all devices undergo preclinical testing and clinical studies to assess safety and efficacy. Marketing authorization includes, as an essential element, evidence that the benefit-risk ratio is beneficial and justifies the use of the device in clinical practice. Nevertheless, the market launch phase and large-scale use exposes a much larger and more heterogeneous population of patients and users, so it is reasonable to expect that postmarket monitoring may highlight new risks or events not previously observed.
UNDERREPORTING: THE PROBLEM OF UNDERREPORTING
The vigilance system is aimed at identifying these events and taking appropriate action, to protect patients and maintain safety. The system is based on voluntary reporting by operators using the devices. It has been reported, however, that there is a phenomenon of underreporting, which undermines the entire surveillance system.
The problem is recognized worldwide. Even in the United States, a recent study published in JAMA Internal Medicine in 2021 reported that 23 percent of events reported as serious injury or death were not correctly attributed to a defective medical device and were reported incorrectly. This was equivalent to about 31,500 deaths caused by unsafe medical devices, which were potentially preventable through an appropriate system of corrective and preventive actions.
Some research in the US, UK, Australia and Canada has investigated the reasons for this problem. Health professionals report their own fear of being blamed, the belief that the errors that occurred were unavoidable and that it was unnecessary to report them, the tendency to avoid bureaucracy, lack of time, lack of knowledge about what to report and how, as well as cultural aspects. Other concausal elements were identified in organizational factors such as inadequate feedback, lack of effective processes and reporting systems.
THE SYSTEM OF “MEDICAL DEVICE VIGILANCE” (MDV)
The device-vigilance system encompasses all activities that aim to increase the health protection and safety of patients and users. The achievement of this goal is only made possible through the reporting of incidents involving in vitro medical and diagnostic devices by all healthcare professionals, manufacturers, or European authorized representatives.
In Italy, the Ministry of Health, as the competent authority, after receiving the report, proceeds to classify and evaluate the data regarding the incidents, taking appropriate measures regarding the medical device involved in the incident, such as the disclosure of information to prevent other events of the same type.
“The recent March 21 decree makes a significant contribution to the efficiency of the post-marketing vigilance system, aiming to solve the technical and procedural criticalities recorded in the past,” comments Prof. Guerino Fares, “although to say the last word we need to wait for the completion of the regulatory framework with the entry into force of the national implementing legislation of the two European Regulations of 2017, the publication of which is expected in August 2022. Only then will we be able to see whether all the chronic knots that experience has shown have really been untied or not.”
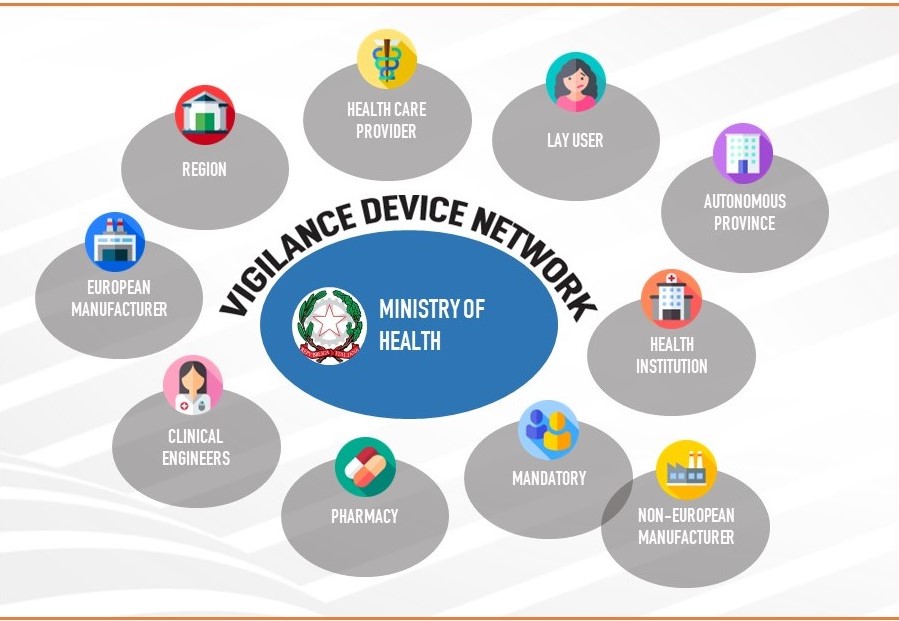
THOSE INVOLVED IN THE MEDICAL DEVICE VIGILANCE NETWORK
The national device-vigilance legislation implements the new European regulations, stipulating that legal representatives of public and private health care facilities and public and private health care providers must immediately notify the Ministry of Health and the manufacturer or its European Authorized Representative of any alteration in the characteristics and performance of a device or any inadequacy in the instructions for use that may cause, or has caused, death or serious deterioration in the health condition of a patient or operator.
In Italy, the national incident information exchange network involves the following:
- Health worker (HOS);
- Local supervisory officer (RLV), in charge of the contact between the health worker and the Regional Supervisory Device Manager;
- Regional Vigilance Manager (RRV), i.e., regional coordinator and contact with the Local Vigilance Manager and the Ministry of Health;
- Ministry of Health (MoH) – Directorate General of Medical Devices and Pharmaceutical Service.
It is the responsibility of the regions and autonomous provinces to ensure the coordination of the device-vigilance network, within the territory under their jurisdiction. Each region is required to identify those responsible for handling incident reports from the public, accredited private and unaccredited private health care system. It therefore falls to the regions to ensure effective synergy between pharmacists, clinical engineers and all other figures involved in the process, both within and outside of health care institutions, as well as coordination with the clinical risk management service.
HOW INCIDENT REPORTING WILL TAKE PLACE
The national network covers the exchange of information on incidents medical devices, in vitro diagnostic medical devices, and devices. the entities named before, will feed the network through an information system implemented in the New Health Information System (NSIS).
The integration of device reporting into the NSIS system is an important innovation, indicating how the worlds of devices and drugs are increasingly converging, thanks in part to the digitization of healthcare systems. This initiative has its origin in the 2019-2021 health pact referred to in the new decree, and in particular pharmaceutical and medical device governance.
In fact, the NSIS system represents the reference tool for measures of quality, efficiency and appropriateness of our National Health Service (NHS), through the availability of information that for completeness, consistency and timeliness, support the Regions and the Ministry in the exercise of their functions and, in particular, the Ministry in its function of guarantor of the uniform application of the Essential Levels of Care (Livelli Essenziali di Assistenza (LEA) on the national territory. So far, the NSIS system contains a section on pharmaceutical services. Soon, the NSIS system will also collect medical device incident reports.
The new reporting system via NSIS will replace the “online incident reporting forms” currently used for both medical devices and in vitro diagnostics.
TIMELINES FOR DEVICE-VIGILANCE REPORTS.
Article 5 and the annex of the ministerial decree March 31, 2022, provide the technical details for sending reports, the final feed to the information system to be borne by the regions.
The device-vigilance system will collect incident reports (both serious and non-serious) from health care providers, manufacturer/device agent reports, and safety actions (see infographic: glossary).
The maximum time for operators to report a serious incident must be no more than 10 days from when they became aware of the event; the deadline for validation of the report by the local device-vigilance manager (RLV), is 3 working days starting from the notification.
“The national device-vigilance network is a circuit fueled by the valuable contribution of all network actors and aimed at encouraging the adoption of every useful measure for the purpose, including the profitable performance by the Ministry of risk management activities. – Prof. Guerino Fares adds. – I am thinking, for example, of the advisability of reinforcing, making it more effective, the obligation of public or private health workers who detect a serious incident to notify the Ministry of Health, in the terms and manner that the competent authority itself should establish in a clear and enforceable manner. This obligation would make the task of reporting easier, which health care workers, lay users and patients who detect an incident, regardless of its severity, may also have to fulfill. I’m thinking, again, of the need to promptly inform the manufacturer of the reported serious incident, who, in turn, in case he has received notice of a complaint from an operator, will dutifully inform the Ministry of Health.”
For further information, reference can be made to the Ministry of Health’s circular dated July 8, 2021, titled “Vigilance of Medical Devices – Guidance for Reporting Incidents Occurring After Marketing in the Light of Articles 87, 88, 89 and 90 of Regulation (EU) No. 745/2017.”
THE ROLE OF THE MANUFACTURER AND THE EUROPEAN REPRESENTATIVE
The manufacturer or its European authorized representative, as the entity legally responsible for the medical devices and in vitro diagnostic medical devices it places on the market, is required to report incidents involving its device to the Ministry of Health. If there is evidence of a possible hazard or risk associated with the use of its devices, the manufacturer is required to voluntarily take preventive and/or corrective action, commensurate with the severity of the hazard, which may result in the disclosure of new safety information, safety instructions, or the withdrawal or recall of the device from the market.
Field Safety Corrective Action (FSCA) are all those measures taken by the manufacturer in order to minimize the future risk associated with the use of a device. This information is communicated to the Ministry of Health and health professionals and constitutes the Field Safety Notices (FSNs).
The manufacturer is required to prepare Field Safety Notices (FSNs) for use by users for all member countries, describing the reasons for corrective action, device inadequacies, risks associated with use, and measures to be taken. The manufacturer must ensure that the organizations involved are reached by the transmission of notices and must require recipients to inform all those who may be affected by the notice.
The Ministry supports the dissemination of this information by posting the safety alerts on its portal, on a dedicated page within the medical device subject area.
Incident reports must be reported by manufacturers/device manufacturers using the Manufacturer Incident Report (MIR Form), which is available on the European Commission’s website and the use of which became mandatory on January 1, 2020. The safety alert forms to be used by manufacturers are those attached to the MEDDEV 2.12-1 rev.8 guidelines.
THE AUTHORIZED EUROPEAN REPRESENTATIVE
The choice of agent (authorized representative or authorized representative) is very tricky for manufacturers that are not based in Europe, as these companies can support the process of reporting and evaluating events through the advice of specialized experts, similar to what is done in the pharmacovigilance sector. This outsourcing approach can be particularly relevant for small and medium-sized companies that often lack dedicated in-house resources or continuous updates on regulatory changes in different countries.
As the new Regulations 2017/745 and 2017/746 state, for manufacturers based outside the European Union, the authorized representative plays a key role in ensuring the compliance of the devices they manufacture and in serving as the contact person established in the Union. The authorized representative is liable before the law for defective devices in the event that a manufacturer fails to comply with its general obligations.
Today, the role of the authorized representative assumes great significance for the entire device life cycle, both in the pre-market trials phase and in the marketing phase. In fact, the responsibilities of defective products also apply to the authorized representative and, as a result, the authorized representative is jointly and severally liable with the importer and the manufacturer. Given the role of proxies, it is appropriate to clearly define the minimum requirements that they should meet, including having a person meeting the requirements applicable to the person responsible for compliance (PRRC).
If the manufacturer anticipates the need for support in the area of incident surveillance, however, it is imperative to accurately qualify the authorized representative, as the regulations do not contain explicitly defined requirements regarding the need for a person with device-vigilance skills, as is the case in the pharmaceutical world (QPPV).
HOW TO PREPARE
With the entry into force of Regulation (EU) 2017/745 (MDR) and 2017/746 (IVDR), the regulatory framework of reference in the field of vigilance for in vitro medical and diagnostic devices, represented by more than 20 years of European directives transposed in Italy by legislative decrees, undergoes a profound strengthening aimed at the development of clear and transparent procedures, suitable for maintaining a high level of safety, while promoting technological innovation.
“It is useful to emphasize the importance of encouraging the adoption by the regions of rational and cogent regulations that provide for the presence of supervisory referents,” concluded Prof. Guerino Fares. – No less significant, on the other hand, is the promotional role that the Ministry of Health should be able to play, organizing awareness campaigns aimed at encouraging the reporting of incidents. I am thinking, finally, of the language in which the manufacturer or his authorized representative will be required to effectively and appropriately draft the safety notice to be provided to the same Ministry.”
To operate in this new environment, it is crucial for manufacturing firms to have access to not only clinical but also regulatory expertise, including through the support of the European representative, in order to be able to interact effectively with the various European authorities and be able to comply with national requirements as well.
In this rapidly evolving system, it is imperative for regulatory authorities to consider the differences in perspectives of the parties involved in recognizing and determining medical device incidents in the context of everyday use. It is essential to train healthcare professionals and lay people, as they are on the front lines of recognizing adverse events, but also to implement a support system at the regional and provincial levels to increase the accuracy and reliability of information on adverse events.








Leave A Comment