Atty. Alessia Nuciari, director of compliance Meditrial.
The in vitro diagnostics industry has undergone a substantial transformation. IVDR Regulation 746/2017, published in May 2017 and applied from May 26, 2022, introduced major changes in the industry. The main changes designate a strengthened system of notified bodies that for the first time will be assessing compliance for about 70 percent of in vitro diagnostic devices. This radical change involves not only manufacturers of in vitro diagnostic medical devices but also investigators and sponsors involved in conducting the clinical studies required by the new regulations.
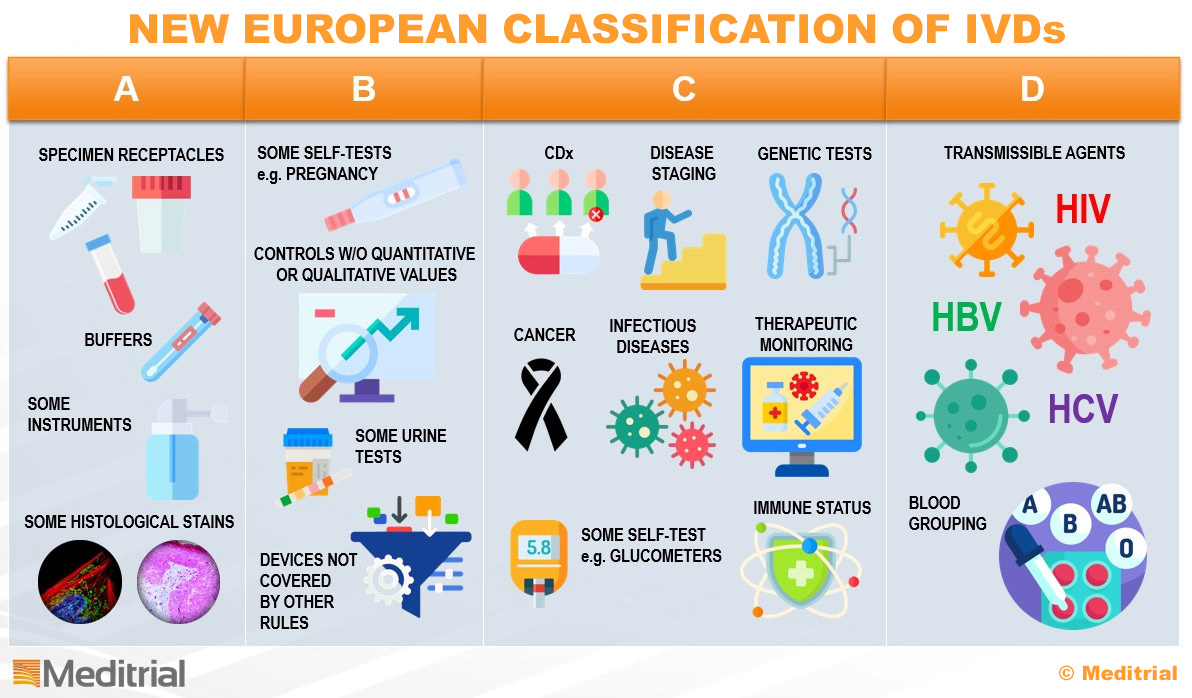
TYPES OF IN-VITRO DIAGNOSTICS
In vitro diagnostic medical devices (IVDs) perform their function outside the body on samples that are taken from the human body. Human subjects are generally not exposed to risk during performance testing of IVD medical devices, except for the risk associated with sample collection procedures or when the information obtained is used for patient management.
IVDs include tests and devices such as test strips and reagents, which use samples such as blood, tissue, or urine, to perform screening, diagnosis, prognosis, predictive testing, and condition monitoring.
Samples are obtained through normal bodily functions or through the use of invasive medical devices that allow the sample to be obtained (e.g., biopsy). Samples are never reintroduced into the human body. These differences make the performance and risk characteristics of IVDs different and unique from other medical devices.
PERFORMANCE EVALUATION
Similar to what is required for medical devices, in the IVD world we have also seen, with the new IVDR regulation, a clearer and more effective illustration of the clinical evidence generation process called “Performance Evaluation” (Annex XIII, IVDR).
It is important to emphasize that performance evaluation should be viewed as an ongoing process necessary not only to generate but also to maintain the clinical evidence needed to support the intended purpose of the in vitro diagnostic device. Therefore, IVDR requires the adoption of a life-cycle approach, whereby clinical evidence must be updated throughout the life cycle of the IVD. Scientific developments and improvements in the state of the art must be reviewed and evaluated by the manufacturer as part of its ongoing and proactive post-marketing surveillance activities.
This pathway includes several steps, where the inputs are the general safety and performance requirements and intended use, and the outputs are the clinical evidence collected during the clinical performance study, explicated in the Final Performance Evaluation Report.
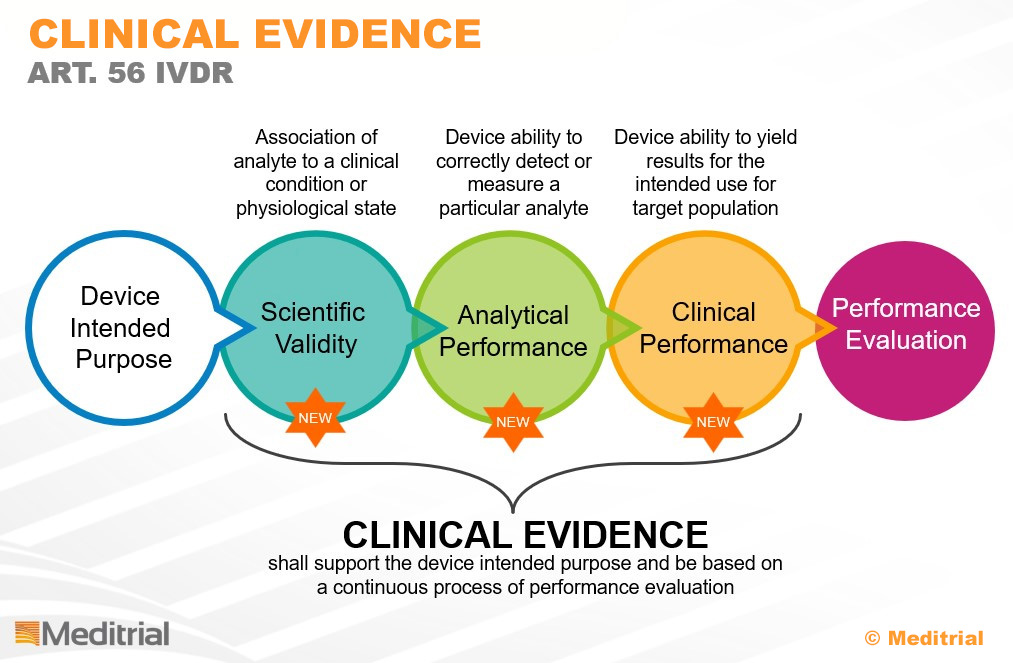
THE PILLARS OF CLINICAL EVIDENCE
As with medical devices, the purpose of the clinical performance study is to capture clinical data necessary to provide the clinical evidence needed to support the compliance of the device under investigation for marketing.
The clinical evidence generated by the study supports “scientific validity,” one of the three pillars of the clinical performance evaluation process-.
Analytical and clinical performance reports, the second and third pillars of the above process, respectively, must also be produced for each device, which flow together with the scientific validity report into the final Performance evaluation report.
In determining what data are needed to demonstrate the safety and performance of IVDs, it is important to consider the existing data available and how to fill any gaps. Where data are not available in sufficient quantity or quality, it will be necessary to generate them. The Clinical performance study with respect to the other two pillars appears to be critical precisely when there is an absence of clinical evidence from the literature and or other studies
THE NEW GCP
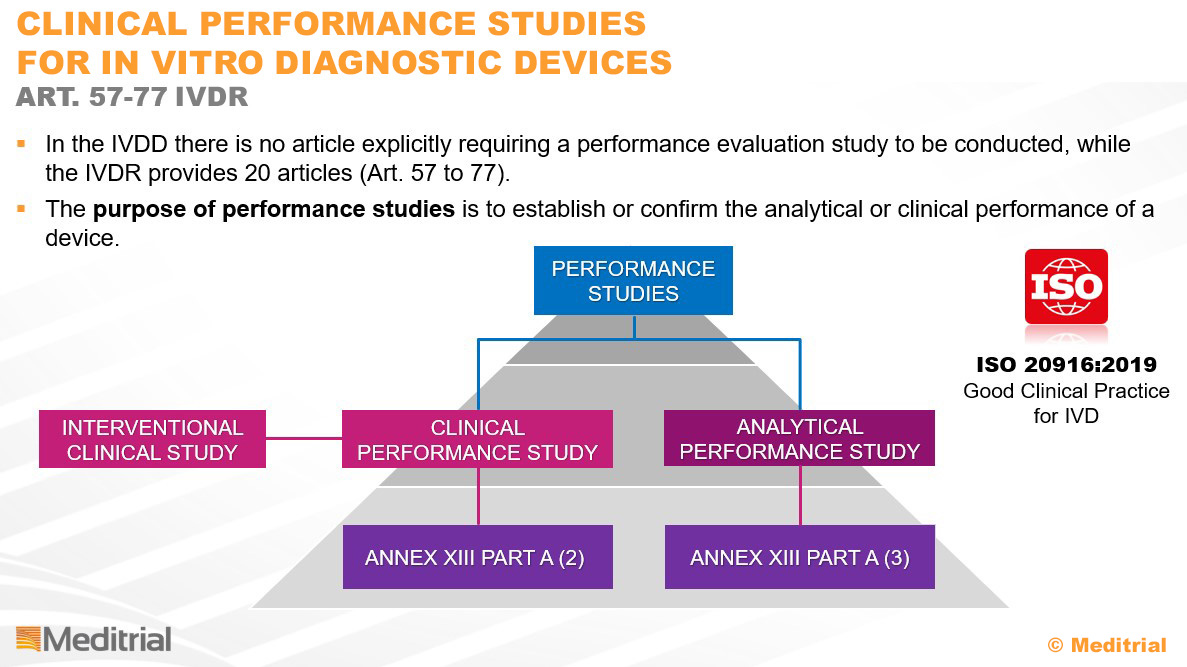
MDCG Guideline 2022-2, published by the Medical Device Coordination Group, in Chapter 6.7 on clinical performance studies identifies among various international guidelines the ISO 20916 standard to support the sponsor in conducting the clinical study in accordance with the mandatory requirements.
ISO 20916 “In vitro diagnostic medical devices – Clinical performance studies using specimens from human subjects – Good study practice” defines good practices for the study, planning, design, conduct, recording and reporting of clinical performance studies performed to evaluate the clinical performance of in vitro diagnostic medical devices (IVDs). This standard is very similar to the harmonized standard for medical device investigations, ISO 14155.
HOW TO CONDUCT CLINICAL TRIALS
Clinical performance studies should always be designed to specify the clinical evidence that the study is intended to produce taking into account potential risks, appropriate ethical requirements, and ensuring compliance with all applicable legal and regulatory requirements.
Through the Clinical Performance Study Plan (CPSP), the criteria, objectives, design, proposed analysis, methodology, monitoring conduct, and registration of the clinical performance study are defined (Annex XIII IVDR 2.3.2).
Clinical performance studies must always be designed to specify the clinical evidence the study is intended to generate taking into account potential risks, appropriate ethical requirements, and ensuring compliance with all applicable legal and regulatory requirements. Because clinical performance studies must be tailored to the specified intended population, the manufacturer must evaluate and justify the use of any sample within its performance study in consideration of the intended purpose and type of IVD. The substantial difference between the world of medical devices and in vitro diagnostic medical devices is the concept of “clinical performance” present only in the latter. The clinical performance of an in vitro diagnostic is its ability to produce results that are closely related to a particular clinical, psychological or pathological condition depending on its intended use (the ability of a test to detect positivity/negativity in a patient).
Atty. Alessia Nuciari, director in charge of compliance Meditrial







Leave A Comment