Research and innovation are based on rigorous clinical trials involving patients firsthand. The main actor in clinical trials is the patient affected by the disease under examination. Including the patient’s perspective allows us to reduce barriers that otherwise limit your participation and to introduce tools and support that provide patients with the best possible experience. We talk about it with Prof. Salvatore Di Somma, director of Emergency-Urgency Medicine at the La Sapienza University of Rome and scientific director of the Italian Association of Patients with Heart Failure (Aisc).
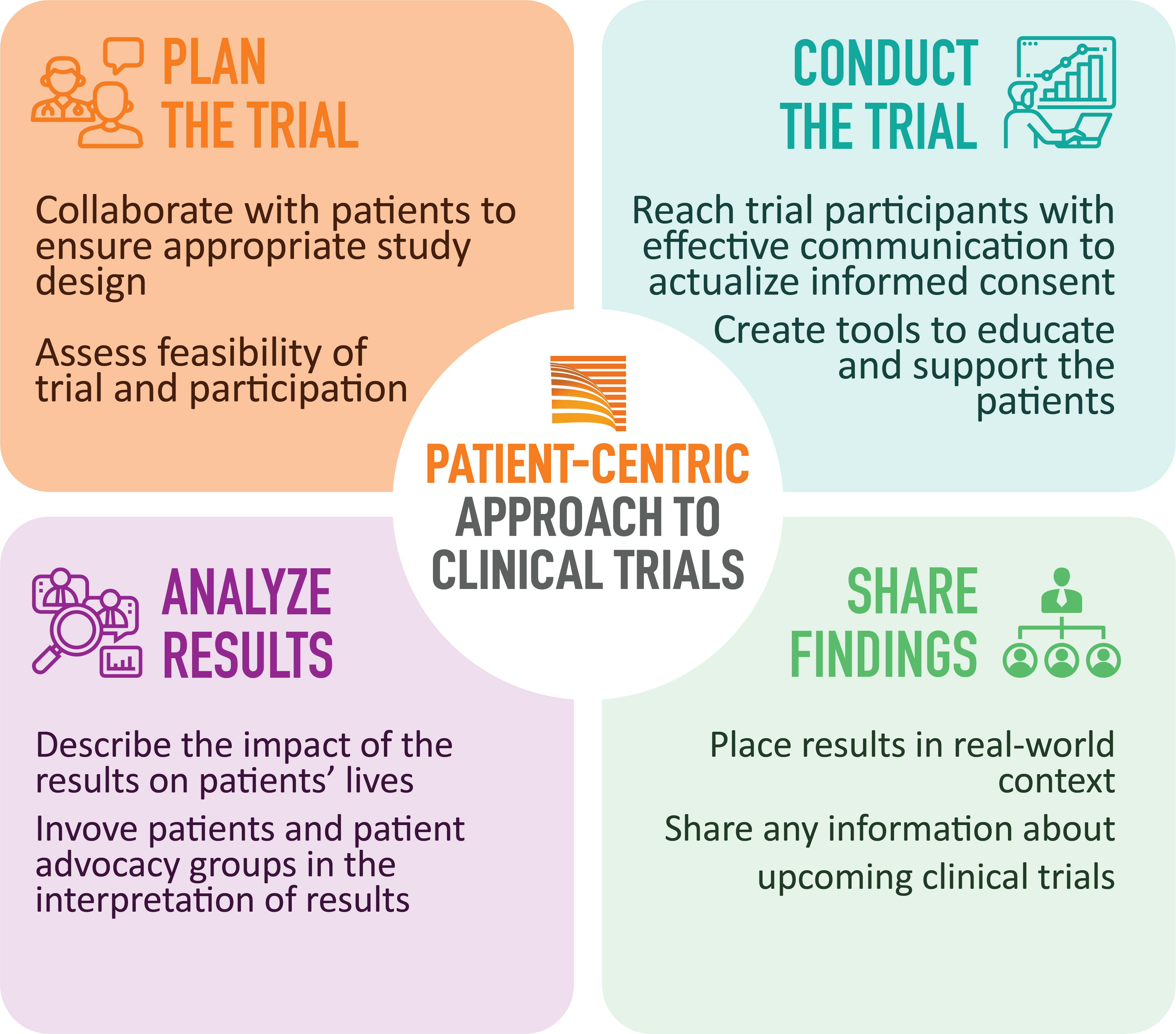
WHAT DOES THE PATIENT THINK ABOUT THIS?
In general, patients are eager to be part of a clinical trial. A 2019 survey found that 79% of people who have never participated in a trial are open to the opportunity. The majority (81%) want to know all the risks and benefits before agreeing to participate, 83% expect access to their data, and 52% want reminders of appointments, calendars, and other interactions to help them manage the experience.
Organizations are realizing the crucial role that patient-centeredness plays in the success of clinical trials. Many companies talk about the importance of being patient-centered, but few have an actionable strategy in their clinical development program. A patient’s life a does not begin and end with his or her involvement in the study: each has needs, concerns, and work, family, and personal commitments that can be far more important than his or her participation. If patients are to continue in the study even when they encounter difficulties or inconveniences, or even agree to participate, it is imperative that the study be designed a priori to accommodate the needs of each participant.
THE PATIENT IN DECENTRALIZED TRIALS
Traditionally conducted clinical trials have seen many of their practices changed because of the Covid-19 pandemic. Before the pandemic they were conducted exclusively in Hospitals or Research Centers. During the pandemic, drug manufacturers faced the prospect of seeing a halt to expensive clinical trials as the pandemic prevented study patients from accessing hospital facilities. In an effort to keep their drug development programs running, biopharmaceutical companies modified traditional protocols to adopt a decentralized form of studies.
To execute decentralized strategies allows patients to participate in clinical trials from their homes by providing them with investigational drugs and treatments where they live.
In addition to the pivotal role played by decentralized trials during the pandemic, these patient-centered studies are gaining praise because of the benefits they provide to patients, research sites, and sponsors:
- Patients prefer these studies because they have access to new therapies and personalized treatments without leaving home.
- Research sites are satisfied because decentralized studies make it easier for them to recruit study patients.
Thus, the role and value of the patient in clinical research have further grown and been recognized. In addition, the pandemic has strengthened the acceptability of studies among patients. Data show that patients like the idea of decentralized studies as a clinical research option because they remove much of the burden of trial participation. Patients have become accustomed to digital interactions with health care providers during lockdowns and expect these tools to be part of future interactions, such as the ability to choose when and where to show up for an appointment via apps and online platforms.
Decentralized practices eliminate many of the burdens that commonly cause attrition, including excessive travel and time commitments. However, it is essential that patients feel emotionally connected to the practice. Such connectivity can be achieved through a constellation of small choices that together create an engaging and supportive experience:
- Use technologies familiar to the patient that can be easily integrated into his or her daily routine.
- Allow patients to use their own devices.
- Provide a central online portal for a simplified digital experience.
- Ideally, fully digital patient interactions will take place through a single platform, where patients can access all their electronic consent forms, electronic diaries, telemedicine links, chat features, appointment calendar, alerts and personal data.
- It should be easy for participants to obtain information and interact with the clinical team.
THE NEEDS OF PATIENTS
Although digital practices aim to be more inclusive, they face several barriers, including the digital competency gap, the exclusion of certain tests and procedures, the complexity of administering medications at home, and the need to build new infrastructure (NPJ Digital Medicine 2022). In addition, the need for contact with one’s physicians remains a central aspect of the therapeutic act.
The need to maintain the balance between technology and human interactions requires new models to be compatible with digitization and continuity of care, especially when the patient is participating in a clinical trial. Even in a fully remote study, many patients will still need periodic support from a real person even to help them navigate the technology, answer questions, or provide reassurance. Therefore, decentralized studies only if well designed to answer not only scientific questions but also the needs of patients will be a viable and globally applicable solution.
Such studies must be built around the needs of the patient population, taking into account their mobility, technological capabilities, fears, access to treatment centers, and clinical conditions.
PATIENT ASSOCIATIONS
Pursuing patient-centeredness requires a coordinated and diverse program, rather than single initiatives such as hosting a patient focus group or conducting a survey. It is necessary to collect data from multiple sources to understand patients’ experiences and use them to build a study approach that best supports patients, their caregivers, and families. Data can easily come from interactions with the patient community. Hence an even greater role for patient associations in research and innovation.
Prof. Di Somma reports his experience: “With the Italian Heart Failure Association we are trying to implement a model that involves optimizing the engagement of the patient when not accustomed to the use of digital devices through the help offered by a caregiver or by the Association itself; at the same time, it is necessary to set up tools that are able to intercept every minimal variation in the health status of the heart failure patient, allowing the physician to change the therapy through the use of televisita and, if necessary, a three-way teleconsultation, also involving the general practitioner. In heart failure, this model could become an official way to prevent flare-ups, to improve care by avoiding cardiology visits in attendance or even emergency hospitalizations, and to reassure the patient himself.”
By building relationships with patient associations, we can learn about the lives of patients with a specific disease and hear from real patients and families how we can make their lives easier. Being aware of how new opportunities like this can be harnessed for the benefit of individuals and clinical trials, rather than continuing with established procedures because “that’s the way it’s always been done,” is a key step toward patient-centeredness. “Patient-centeredness is becoming the new paradigm in medical science, both in clinical practice and clinical research. However, centrality means adequate representation, hence the importance of patient associations” reiterates Prof. Di Somma.
Aifa (Italian Medicines Agency) has set up a permanent consultation table with patient associations for constant and active involvement of all citizens on drug-related issues: a valuable contribution to regulatory activity because it brings real experiences and expertise that will improve the promotion and protection of citizens’ health.
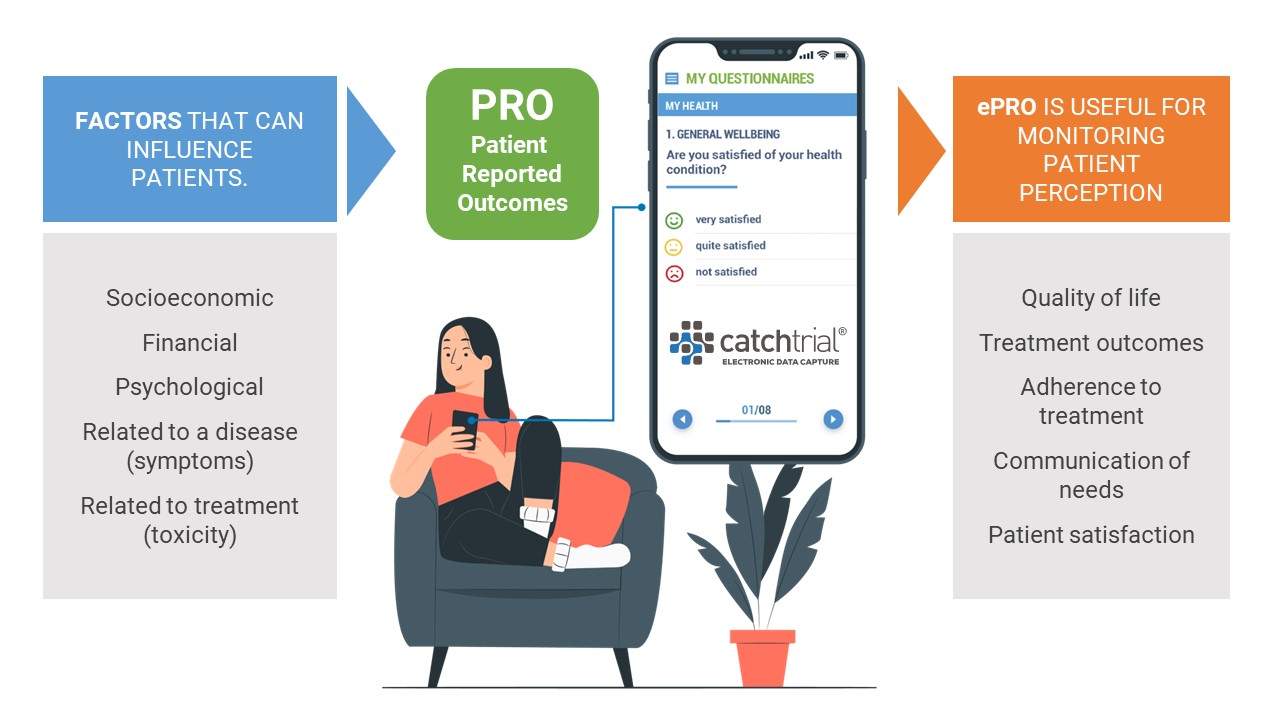
PROs, HEALTH INDICATORS REPORTED BY PATIENTS
Thanks to advances in technology, dissemination of information, and associationism, patients today are increasingly willing and eager to actively participate. The English term PROs (patient-reported outcomes) represents “a report on a patient’s health status that comes directly from the patient, without interpretation by a physician or anyone else” (Guidance for Industry Patient-Reported Outcome Measures).
PROs can refer to disease symptoms; treatment side effects, such as pain, fatigue or anxiety; functional outcomes, such as physical, sexual or functional outcomes, such as physical, sexual, social, role, emotional or cognitive functioning; or multidimensional constructs such as health-related quality of life (HRQOL) or health utility.
PROs can be included as clinical trial endpoints and administered through validated questionnaires with a standardized assessment approach. PROs can be administered by different modalities, from the most traditional (e.g., paper questionnaire, interview, telephone) to the most innovative, which are based on E-PROs technologies.
THE PATIENT EXPERIENCE
The Eupati is a European program created in 2012 with the aim of educating patients and actively involving them in the process of drug research, testing and development for constant and productive discussion with decision-making bodies. In 2019, thanks to Accademia dei Pazienti, a new professional figure in the world of healthcare has also emerged in Italy: the expert patient, certified Eupati.
The expert patient acts as spokesperson for the needs of all patients, bringing with him the baggage of their “real” experiences, lived in contact with pathology. Therefore, he can competently represent them during the institutional events in which he participates, dialoguing “up close” with institutions, ethics committees, associations of doctors and nurses, and all those who work in the medical-health field. Currently in Italy, two ethics committees-the one at San Raffaele in Milan and the one at Gemelli in Rome-have chosen an experienced Eupati patient as their internal member. But since there is no law, even in the current reform of ethics committees, this participation still depends on the sensitivity of those who order the internal members of their ethics committees.
AMPLIFYING THE PATIENT’S VOICE
Future research will be designed to structure innovative technology and design to meet patients’ de needs. By involving patients and their family members and/or caregivers from the very beginning of clinical trial design and considering how new digital technologies can best be used, it is possible to create studies that attract and retain more and more participants and operate more efficiently.
To improve the patient experience while participating in a clinical trial as a whole, we must consider every aspect of care, including communication and the environment. Efficient processes that deliver the required outcomes are key components of a clinical trial, but they alone are not enough to ensure an excellent patient experience. Instead, it is necessary to involve patients in all processes that impact them and ensure that their experience of participation is fully understood. With this approach, trials will have the best chance of producing positive outcomes for everyone.

 In this regard,
In this regard, 





Leave A Comment