Edited by Dr. Monica Tocchi, Meditrial medical director.
The organization called ICH (International council for harmonization of technical requirements for pharmaceuticals for human use) is a global organization that unites regulatory authorities and the pharmaceutical industry in the common goal of ensuring the development of high-quality medicines. The ICH publishes numerous guidelines used internationally for the clinical development of pharmaceuticals.
CLINICAL TRIAL DESIGN AND STATISTICS
Guidelines marked with the letter E, its initials for Efficacy, cover the design of clinical trials, the design, conduct, safety and reporting of clinical trials, but also new types of medicines derived from biotechnological processes and the use of pharmacogenetics/genomics techniques to produce more targeted medicines. Among these, Guideline E9 reports statistical principles for clinical trials.
In November 2019, ICH published an addendum to the aforementioned E9 guideline. The addendum, titled “Addendum on estimands and sensitivity analysis in clinical trials to the guideline on statistical principles for clinical trials,” introduces new statistical concepts destined to revolutionize the future of clinical trials. The document was promptly adopted by both the European Medicines Agency (EMA) and the U.S. FDA.
THE ESSENTIAL TERMS
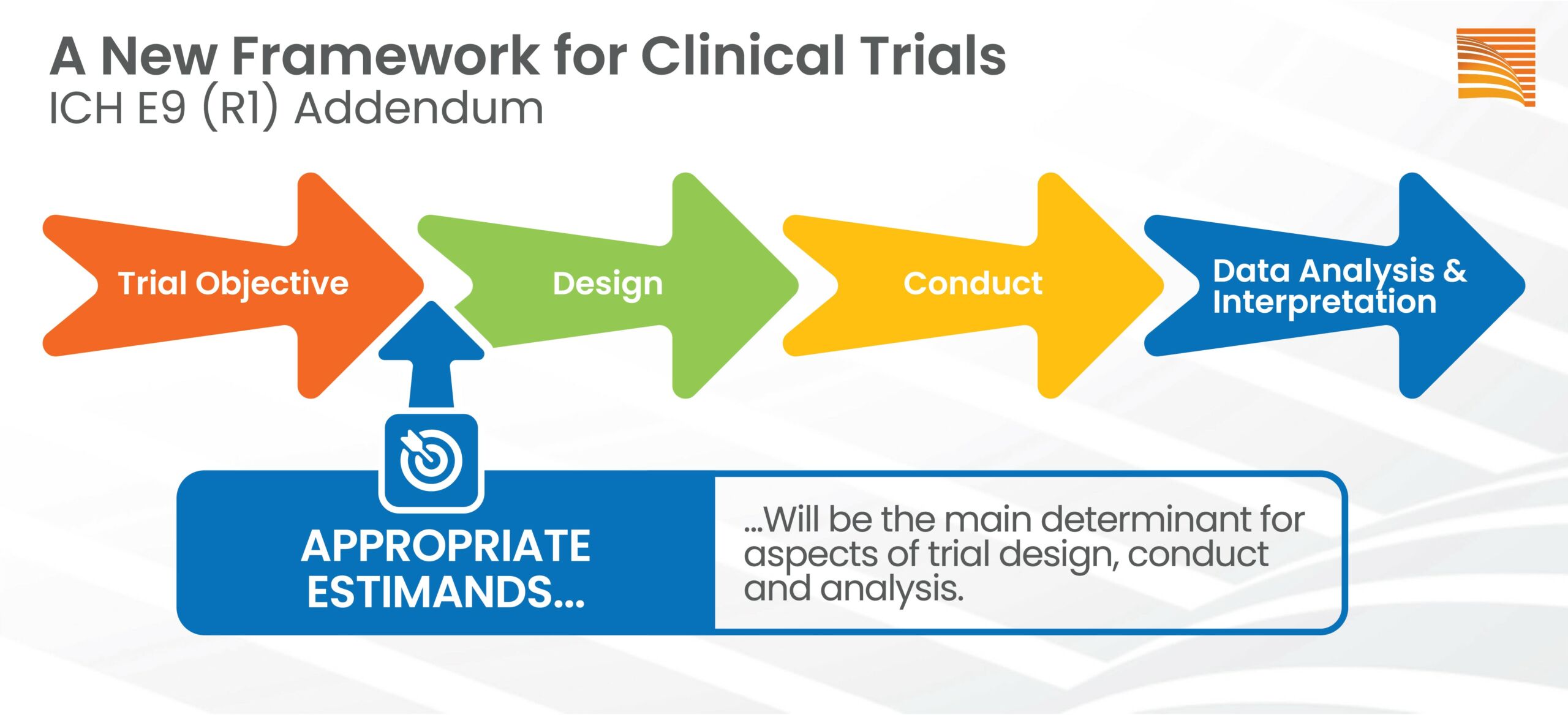
ICH Addendum E9 (R1) represents a useful model for strengthening the dialogue between the sponsor and all stakeholders involved in the formulation of the objectives, design, conduct, analysis, and interpretation of clinical trials, especially with regard to treatment effect(s).
The starting assumption for the new principles introduced by the guideline is the need to clearly describe the risks and benefits of a treatment (drug) for a given disease condition in order to facilitate adequate information to pharmaceutical companies, regulatory authorities, and all stakeholders. In the absence of such clarity, there is a risk that the reported treatment effect may not be correctly interpreted.
The estimand is defined in the guideline as “the objective for answering the scientific question of interest posed by the study objective” and has five attributes: population, treatment, variable (endpoint), intercurrent events (defined as those events that may occur after randomization and that preclude or change the interpretation of the variable) and summary measure. An example of an intercurrent event in the clinical trial might be treatment discontinuation.

STUDY PLANNING
The objective of a clinical study is to answer pre-defined research questions. Goal setting involves the main parameters of the study, such as: subjects to be included, data management (including missing data), and the method of statistical analysis. The ICH represents a framework for estimators[1], which is useful in precisely defining the treatment effect.
Study planning should proceed according to a sequential method. The objective of the trial must determine the choice of estimands, and those estimands must determine the choice of estimators, not the other way around. The construction of the estimand must take into account what is of clinical relevance to the particular treatment in the relevant therapeutic setting. Underlying considerations include:
- Pathology under study;
- Clinical context (e.g., whether alternative treatments are available);
- Type of treatment administration;
- Goal of treatment (e.g., for preventive purposes, or symptom control…).
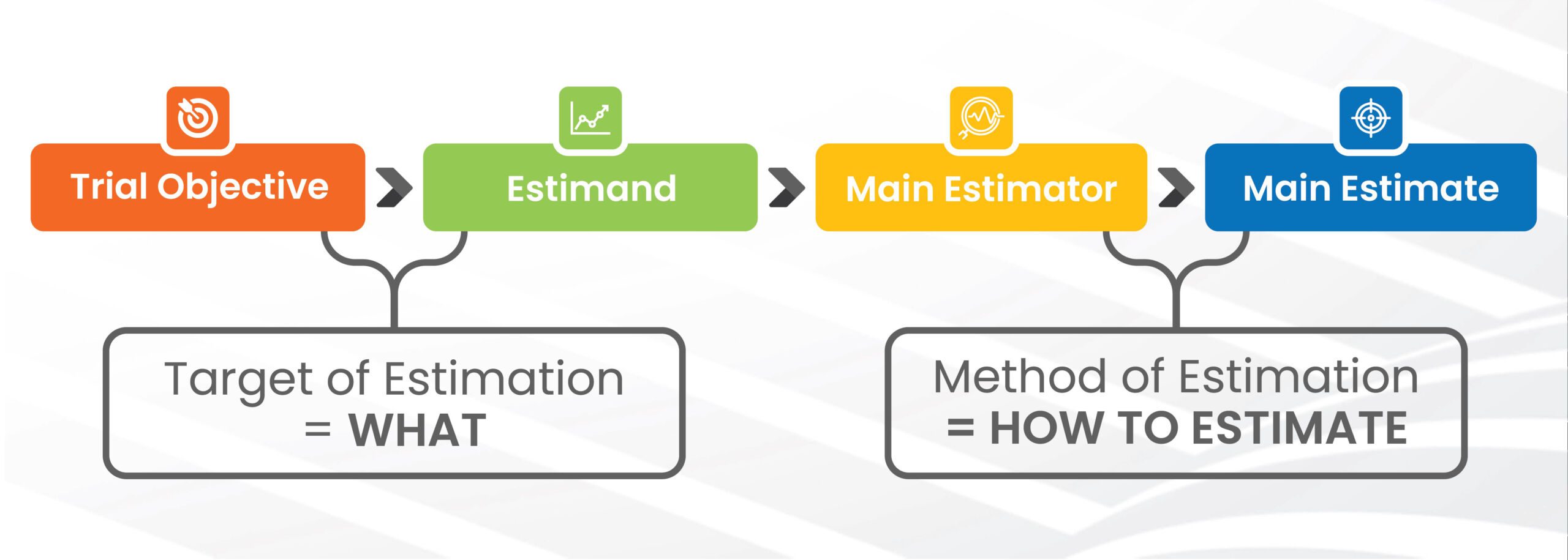
GOAL AND METHOD OF THE ESTIMAND
Each individual investigation objective, or endpoint, must be matched by an appropriate estimand (what is to be estimated). More specifically, the addendum to ICH E9, suggests aligning the estimand using sensitivity analysis, for each given endpoint of the trial. Identifying such cascading steps allows us to have a reliable and more effective statistical methodological approach, but more importantly it allows both Sponsors and regulatory reviewers to communicate on a common substrate.
Defining the study objectives should be an iterative process, directly predicting how the events intercurrent with the treatment effect of interest will be handled.
INTERCURRENT EVENTS AND MISSING DATA
Intercurrent events are events that occur subsequent to the onset of treatment and that affect or may affect both the interpretation and the very reliability of the measurements related to the question under interest. Therefore, it is necessary to consider potential intercurrent events precisely when describing the question under investigation in order to accurately define the treatment effect that is to be estimated.
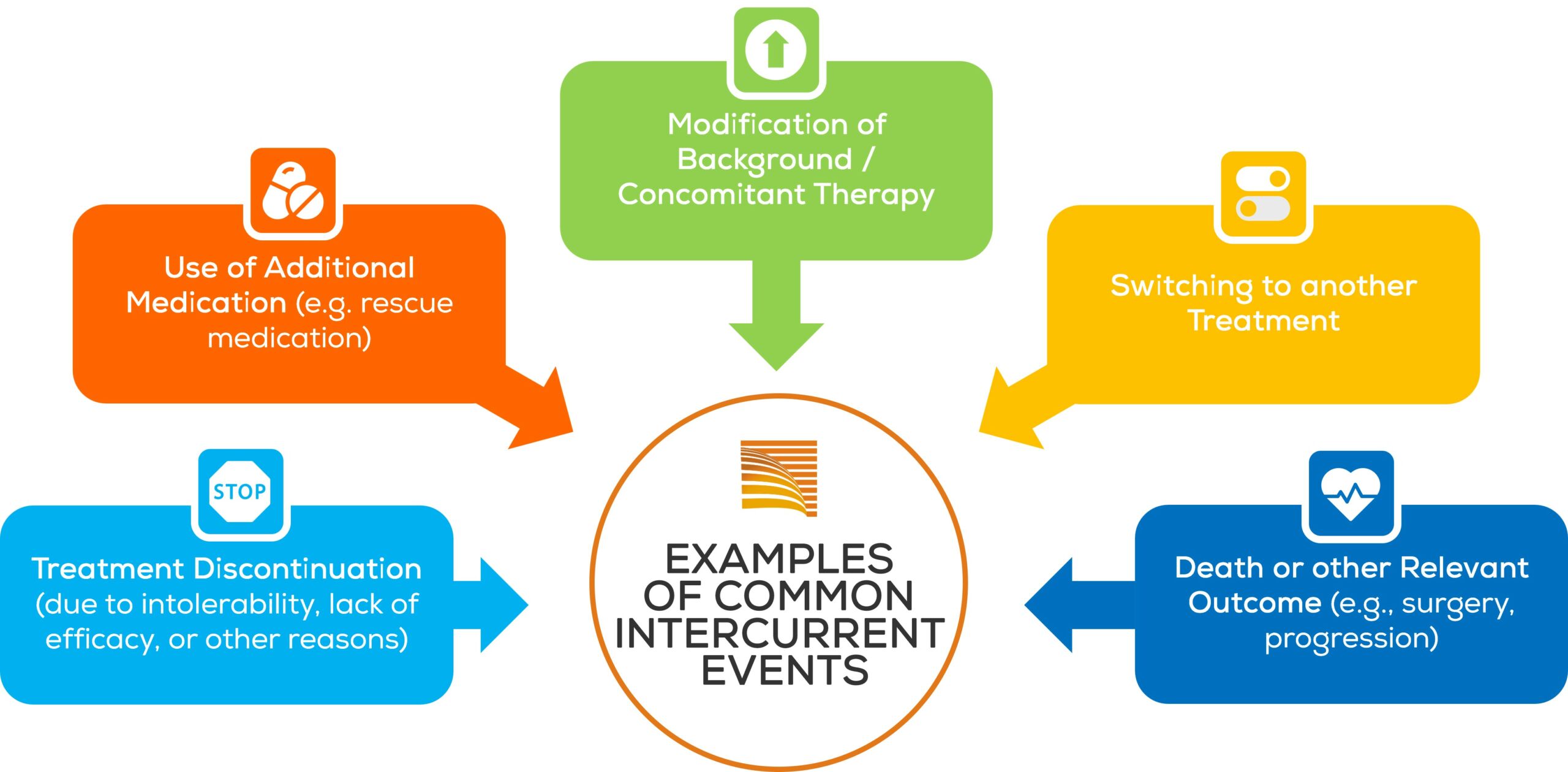
Intercurrent events result from various patient pathways during the course of the study. For example, if we are interested in a patient reported outcome (PRO) endpoint at month 6, some patients will still be on treatment and their PRO results will reflect the 6 months of treatment, others may stop treatment because of an adverse event and start a new therapy.
Taking another example: imagine an end-of-life study in cancer patients, in which drugs are developed and administered to maintain weight, clinical status, and quality of life for the patient’s remaining lifespan.
Thus, measures to assess efficacy will be items such as body composition (DEXA), body weight, hand grip strength, and quality of life (QoL) rating scales. The intervening event here will be that a nonnegligible percentage of patients may not survive and must be considered in defining the treatment effect of interest.
In the absence of an indication of the estimand (effect of the treatment of interest) within the clinical protocol, deaths will be treated as missing data and therefore it will be difficult to interpret the results of the survey; therefore, it is essential to consider missing data in addition to intercurrent events, i.e., those data that would be significant for the analysis of a given estimand but have not been collected, which should be distinguished from data that do not exist or from data that are missing due to an intercurrent event.
THE NEW TRIALS.
The application of the E9 guideline is growing in studies conducted different therapeutic areas. Authorities have already recognized value in providing a common language to facilitate discussions with all stakeholders in clinical trials. For example, the FDA guideline called “Incorporating Clinical Outcome Assessments into Endpoints for Regulatory Decision Making” includes a discussion of estimands and presents an example of application in breast cancer, considering not only clinical variables, but also and especially patient-reported outcomes (PROs).
This new approach to measuring therapeutic effects, linked to study objectives, will radically change the way clinical trials are designed, analyzed and interpreted, as well as the data collection strategy. In the context of PROs (patient reported-outcomes), there is an emerging opportunity to specify more precisely the rationale for patient-centered objectives. By developing precise objectives, especially considering the patient’s perspective of the treatment, and adopting appropriate estimands, it will be possible to ensure the collection of data that possess real clinical significance and provide a clear interpretation. Such an approach enables interpretative uniformity and clear assessment of the effect of patient-centered treatments, contributing to the rapid approval of effective therapies.
Edited by Dr. Monica Tocchi, medical director.
[1] Estimandi (Estimand): The word estimand used here refers to a specific statistical technical term used in clinical research, namely estimand, for which ICH E9 defines the scope and meaning.







Leave A Comment