Dr.ssa Paola Antonini, Meditrial scientific director
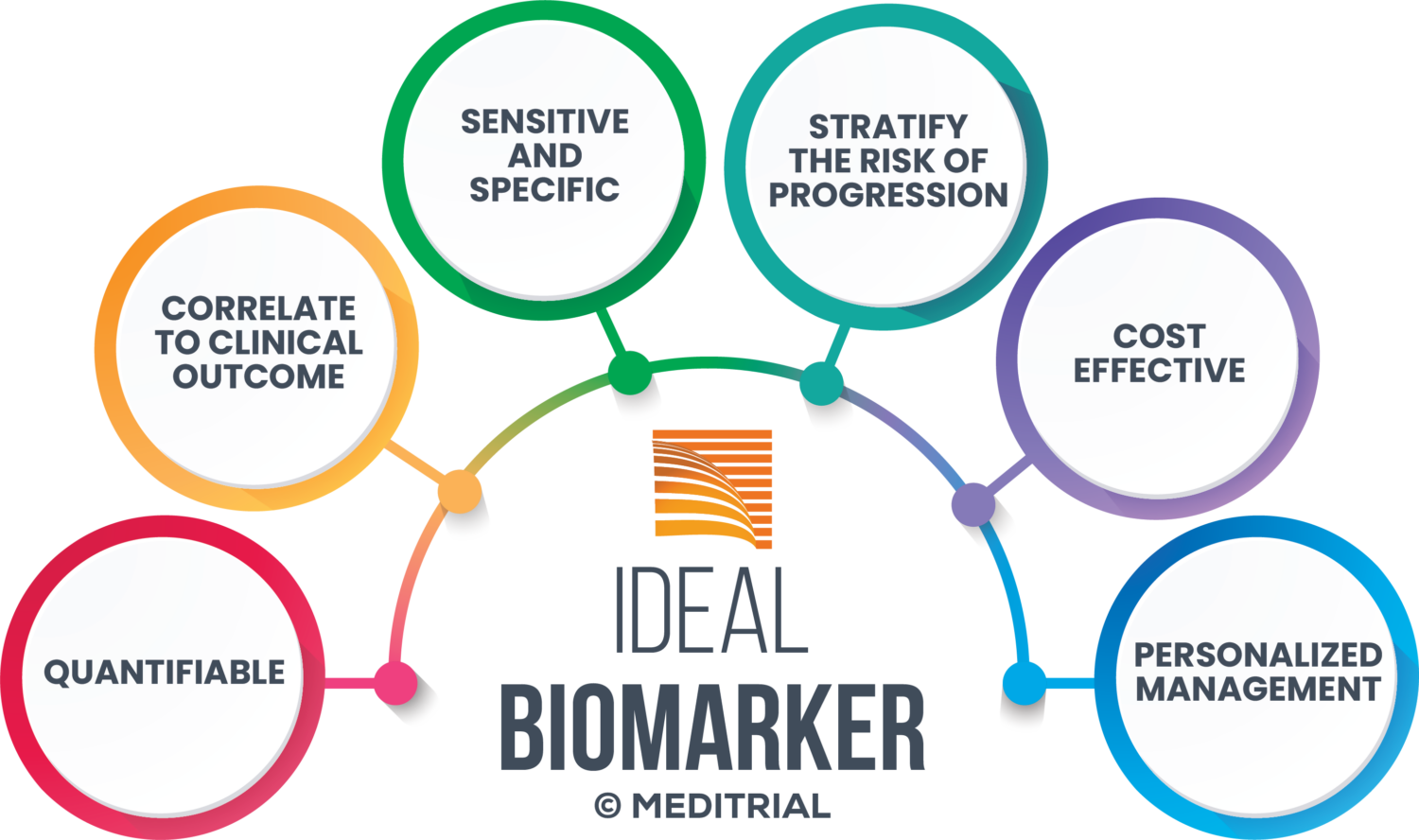
In recent years, the field of biomarkers in research and clinical practice has shown remarkable progress. These tests can be used in the screening, diagnosis, characterization, and monitoring of diseases; as prognostic indicators; for the development of individualized therapeutic interventions; for the prediction and treatment of adverse drug reactions; to identify cell types; and for pharmacodynamic and dose-response studies. The use of clinical biomarkers is easier and less expensive than direct measurement of the final clinical endpoint, and biomarkers are generally measured in a shorter time frame. To understand the value of a biomarker, it is necessary to know the pathophysiologic relationship between the biomarker and the relevant clinical endpoint.
The identification of reliable biomarkers can contribute strongly to the development of new therapies and increase the success rates of clinical trials. By moving to a more personalized study model, protocols can be designed dedicated solely to patients most likely to respond to treatment. In clinical practice, this approach makes it possible to provide personalized, safer and more effective treatments.
This article discusses the latest innovations in the field and why biomarkers will be increasingly relevant in the development of new therapies.
BIOMARKER TYPES
The FDA defines biomarker as a defined and measurable characteristic of normal biological or pathological processes or response to therapeutic intervention. Biomarkers of safety and efficacy can be distinguished.

Biomarkers can consist of DNA, RNA, proteins, metabolites measured from blood, urine or saliva samples, biological fluids and tissues as well as tumor masses. While quantitative biomarkers provide values distributed on a continuous scale, some biomarkers of a qualitative nature provide a binary result (present or absent). The distinction between biomarker-positive and biomarker-negative individuals is made on the basis of a threshold value (or cut-off) applied to quantitative measurements, which is often determined through studies of retrospectively analyzed samples.
BIOMARKER CLASSIFICATION
A classification of biomarkers is provided below:
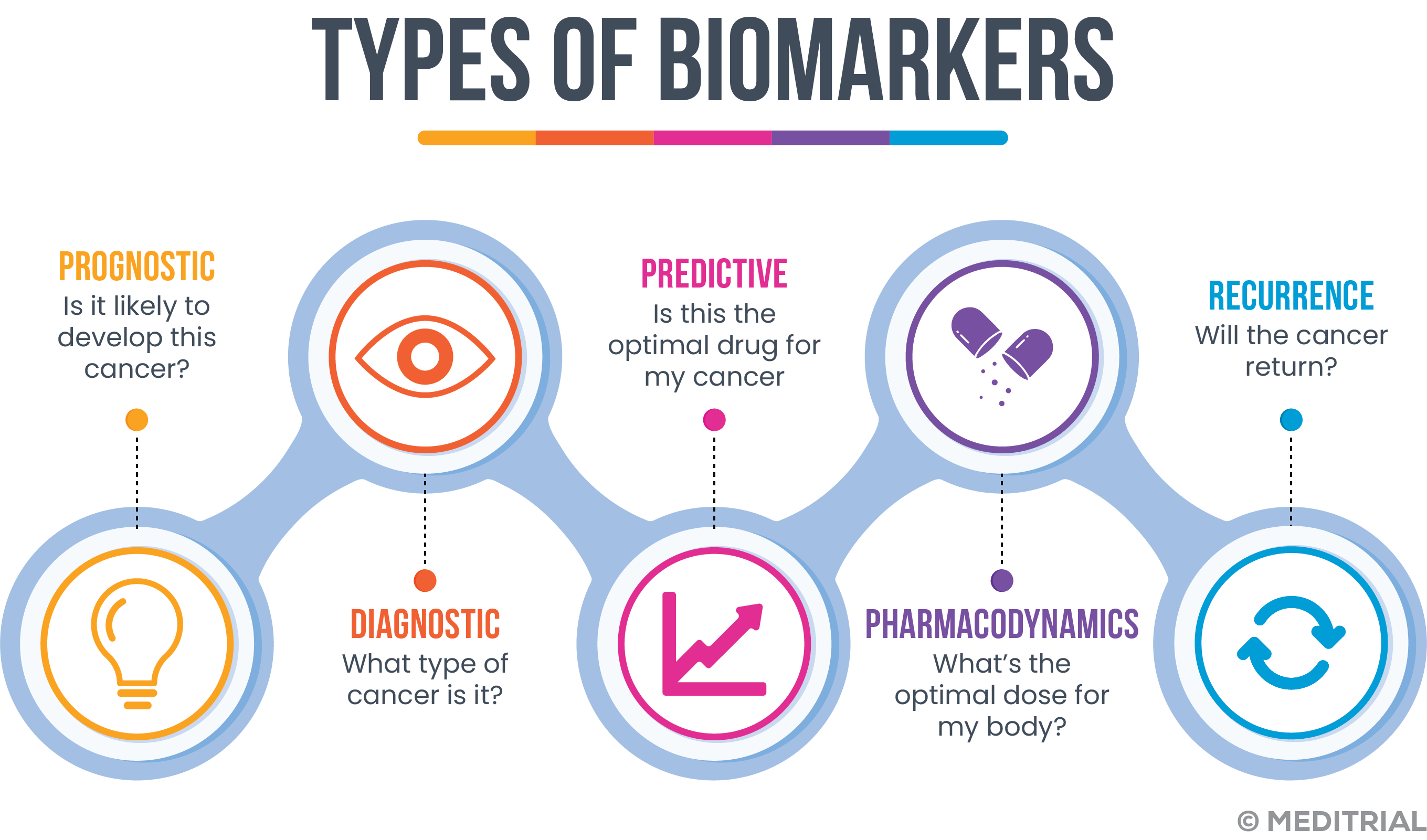
- Prognostic biomarkers: a biomarker is defined as prognostic when it is able to provide information about the history of the disease, regardless of the therapies the patient receives. Prognostic biomarkers are able to indicate the likely outcome of a disease, in the absence of therapeutic intervention. It can be used as an enrichment strategy (Enriched Clinical Trials) to select patients who are likely to have attractive clinical outcomes or progress rapidly.
- Predictive biomarker: A biomarker is predictive when it provides information about the activity of specific therapies thus allowing patients to be stratified according to their likelihood or otherwise of responding to a given treatment. These markers can also be used as an enrichment strategy in clinical trials to identify a subpopulation that may have a favorable or unfavorable response to a treatment.
- Pharmacodynamic biomarkers: These biomarkers help determine the pharmacological effects of a drug and may provide insight into the efficacy of the treatment. Most pharmacodynamic biomarkers, are mainly employed during phase II studies to better understand how to employ a drug and guide in dose selection (Proof-of-concept and dose-ranging studies). They are rarely used as a basis for regulatory approval.
BIOMARKER APPLICATION IN CLINICAL TRIALS
The efficiency of a clinical trial can be greatly enhanced by a protocol that uses biomarkers to stratify patients during enrollment or to monitor treatment response. Biomarkers also improve patient selection for a given clinical trial by allowing for sample reduction.
One benefit biomarkers provide is clinical trial optimization, identifying patients who are more likely to gain benefit from a particular product while excluding patients who may have a higher risk of developing adverse reactions. This latter aspect has important ethical and social implications. Patients negative for the reference biomarker could be excluded from trials with treatments that are ineffective or unsafe for them.
THE DESIGN OF BIOMARKER-BASED STUDIES
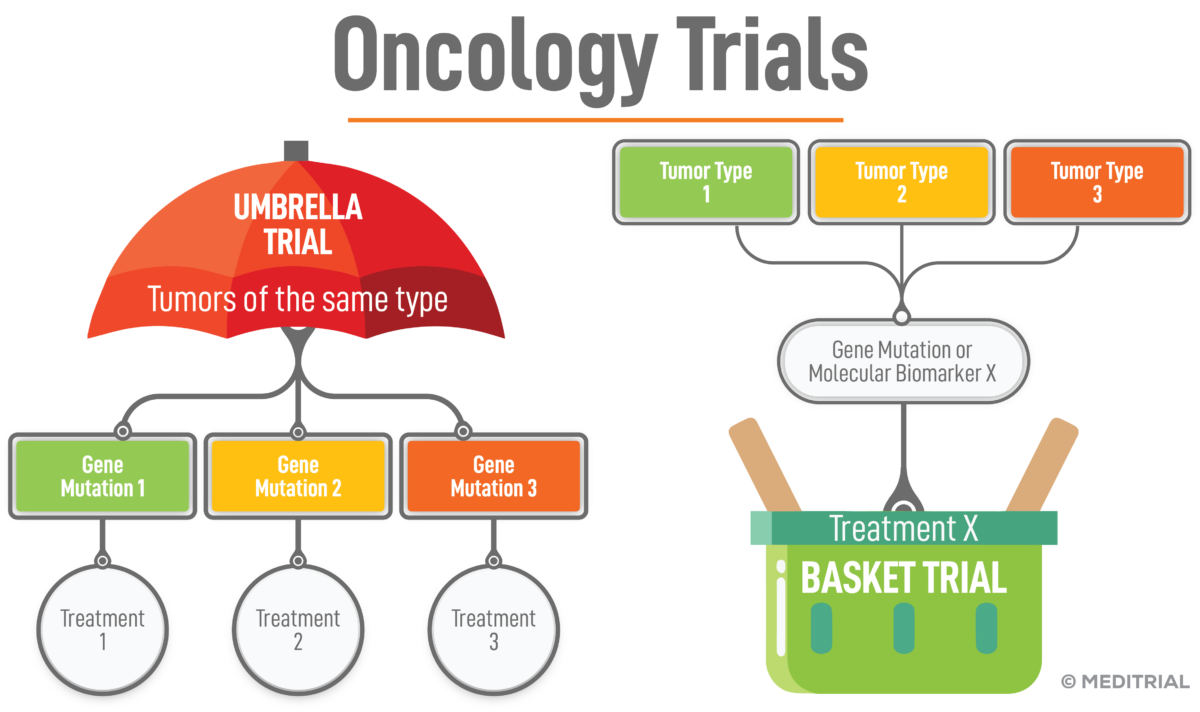
There are several types of biomarker-based clinical trials, including clinical trials involving the combined development of the drug and its biomarker (companion diagnostics) in the context of precision medicine.

A typical design that includes only biomarker-positive patients are basket trials; these trials are performed to test the efficacy of a new drug in patients with different types of a disease (e.g., oncologic disease) but who all have the same mutation, biomarker positive. In basket trials, patients receive treatment targeted at a specific mutation, not a specific tumor.
Another possibility is the “umbrella” trial, in which multiple therapies targeted at different genetic mutations in a single disease (genetic biomarkers) are evaluated. In this type of study, participants are stratified into subgroups based on genetic alterations.
BIOMARKER AS SURROGATE ENDPOINT
Biomarkers can be used as surrogate endpoints in clinical trials, based on the assumption that there is a significant correlation between the value of the biomarker, and a specific clinical endpoint of interest. The advantage of biomarkers is that they can more quickly determine treatment effects, relative to the clinical endpoint of interest. In addition, in some circumstances it is not possible to measure clinical responses directly; in these cases, biomarkers provide an alternative method for evaluating experimental treatment.
Other indirect advantages of using biomarkers as surrogate end-points include reducing trial sample size and enabling researchers to make decisions more quickly. In diseases with poor prognosis, there are important ethical advantages to using biomarkers as surrogate endpoints.
Surrogate endpoints can measure the underlying cause of a disease or be a predictor of the final outcome. Many diseases, however, occur with multiple pathophysiological effects. In this case, biomarker use may yield dubious results and is not useful in predicting treatment efficacy.
In designing a study that includes biomarker as an endpoint to demonstrate the clinical benefit of a therapy, certain conditions need to be verified.
- Is there reliable and consistent epidemiological evidence to support the link between the endpoint and clinical benefit?
- How strong is the correlation between the endpoint and clinical event?
- Has the biomarker been shown to predict clinical benefit using a different drug that has the same mechanism of action?
BIOMARKERS IN ONCOLOGY
Biomarkers represent the fundamental basis of modern cancer therapy. Research has shown that tumors have distinct characteristics, even within the same type of cancer. Cancer biomarkers are now used by physicians to learn more about a patient’s tumor and predict which treatment is most likely to work against each specific cancer. Cancer biomarkers are biological substances produced by the body or tumor of a cancer patient. Biomarker testing helps characterize changes in the tumor.
Biomarkers could be tumor-specific DNA, RNA, protein or metabolomic profiles. Genomic testing examines the DNA sequence. Biomarkers can be used for a variety of purposes, including determining a person’s risk of developing cancer; determining a person’s likelihood of cancer recurrence; predicting the likelihood that a specific therapy will be effective for a specific patient; and monitoring course of a disease to see if a treatment is effective. These biomarker tests are typically genetic in nature, looking for alterations in genes that influence cancer progression.
Many clinical trials are underway in different areas of biomarker discovery. In addition to identifying new targets and determining their significance, new research focuses on several areas.
- Response to immunotherapy: immunotherapy has revolutionized cancer treatment, but not all patients respond the same way. Researchers are identifying biomarkers that predict immune response to assess which patients benefit from which type of immunotherapy.
- Liquid biopsies: liquid biopsies can detect DNA released by tumors, known as circulating tumor DNA. A simple blood draw can reveal common DNA changes to tailor treatment options; it can also assess whether the tumor is responding and investigate how the tumor develops resistance to specific therapies based on additional genetic changes discovered in subsequent blood tests.
- Minimal residual disease: this type of biomarker can be assessed through liquid biopsy testing after therapy has been completed and no identifiable disease is present after examination and imaging. Through blood analysis, minimal residual disease assesses the remaining disease at the molecular level. It could be used to assess which patients would benefit from more aggressive treatment and which could be spared from such treatment.
- Pharmacodynamic markers: use a biopsy before and during treatment to examine dynamic molecular changes within the tumor and assess whether the drug is working as intended.
THE SEARCH FOR NEW BIOMARKERS
The discovery of new biomarkers is now considered a keystone in the future of clinical drug development.
An increasing number of biomarkers are being discovered and identified by preclinical research, but only a few of the many experimental biomarkers, are found to be clinically useful. Clinical validation of a biomarker is established by authenticating its correlation with clinical outcome. This process allows evaluation of performance as well as determination of the range of conditions under which the biomarker will provide reproducible and accurate data.
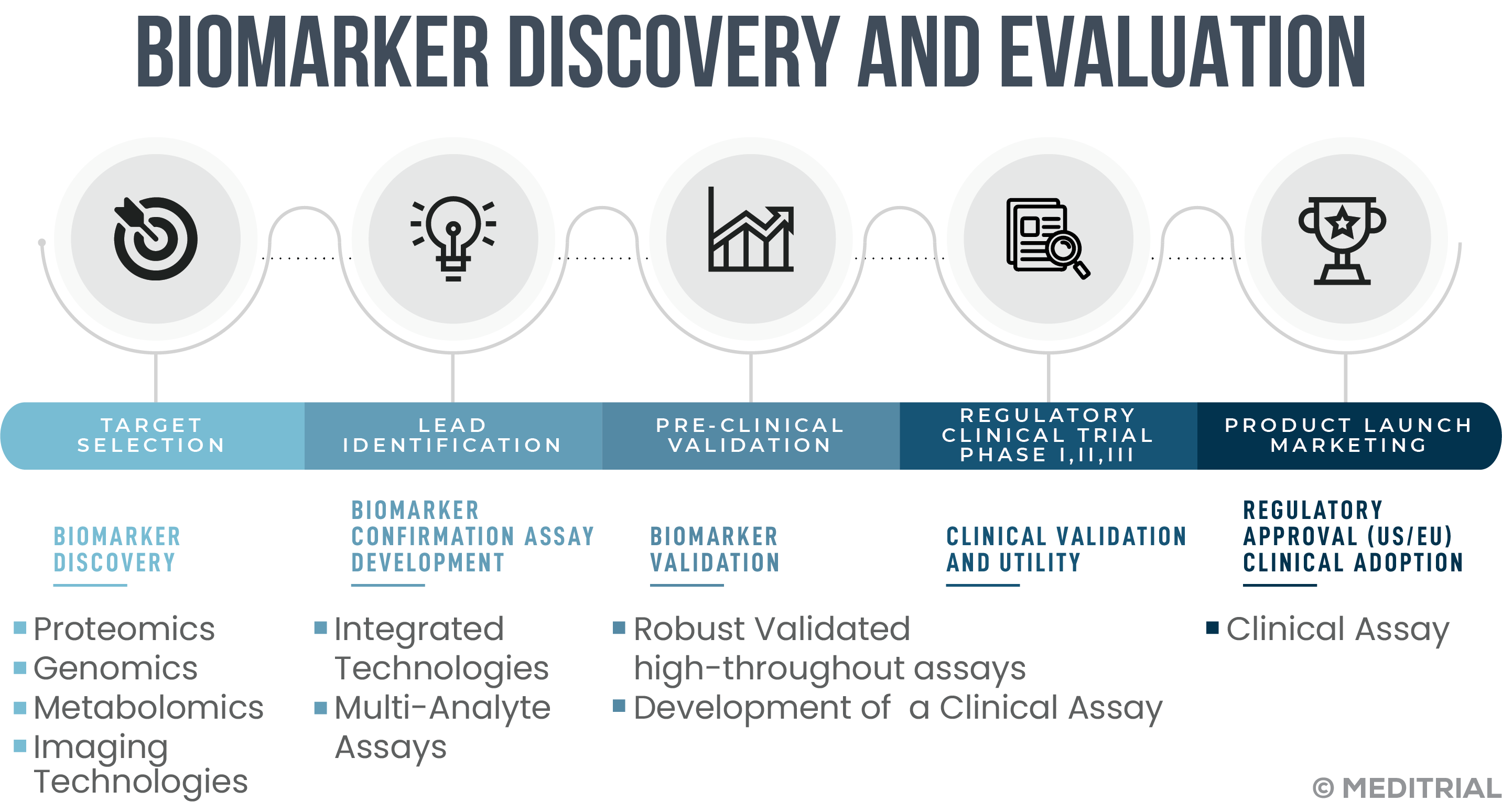
Clinical research data transposed to clinical practice indicate how accuracy in biomarker validation is critical to patient benefit. A false-positive result will expose patients to unnecessary and ineffective treatment, while a false-negative result could result in the discontinuation of a potentially effective treatment.
New methodologies based on genetics, metabolomics, and proteomics make a substantial number of new biomarkers available. Their use in clinical research is an ongoing reality, while their use in clinical practice is still partial. For application in clinical routine, it is necessary to have scientific recommendations for each biomarker, based on robust evidence of correlation and consistency with clinical events in the disease under investigation.
RECOMMENDATIONS FOR CLINICAL RESEARCH
Despite the advantages, the path of clinical trials in combination with biomarkers has some limitations. The first relates to the characterization of pathology into such numerous subtypes (e.g., in oncology) that the diseases themselves are “rare” and thus jeopardize study feasibility. A second major problem concerns the inherent specifics of the biomarker and treatment target. The changing molecular dynamics of both vary over time, in a manner closely related to the stage and type of disease and treatments. For these reasons, a patient who might be categorized as a “nonresponder” based on biomarker today might become eligible in the study at later times as a consequence of the change in the molecular profile of the biomarker and its target.
The success of studies using biomarkers as surrogate endpoints requires multidisciplinary cooperation between different sectors, where CROs with scientific and regulatory expertise, such as Meditrial, have a strategic role in defining and ensuring endpoints that are scientifically valid and in line with the requirements of regulatory authorities.







Leave A Comment