Over the course of two years of the pandemic, there have been heated debates about the safety and efficacy of the anti-Covid-19 vaccines. Even today, after more than 7 billion doses administered, issues continue to emerge about the available clinical evidence (Il Tempo). Reports often reported in the lay press create concern and perplexity in the population. In this article, we used the case of vaccines to examine the mechanisms of generating scientific evidence for medicinal products, current issues, and new opportunities in Europe for better population health protection.
ABSTRACT
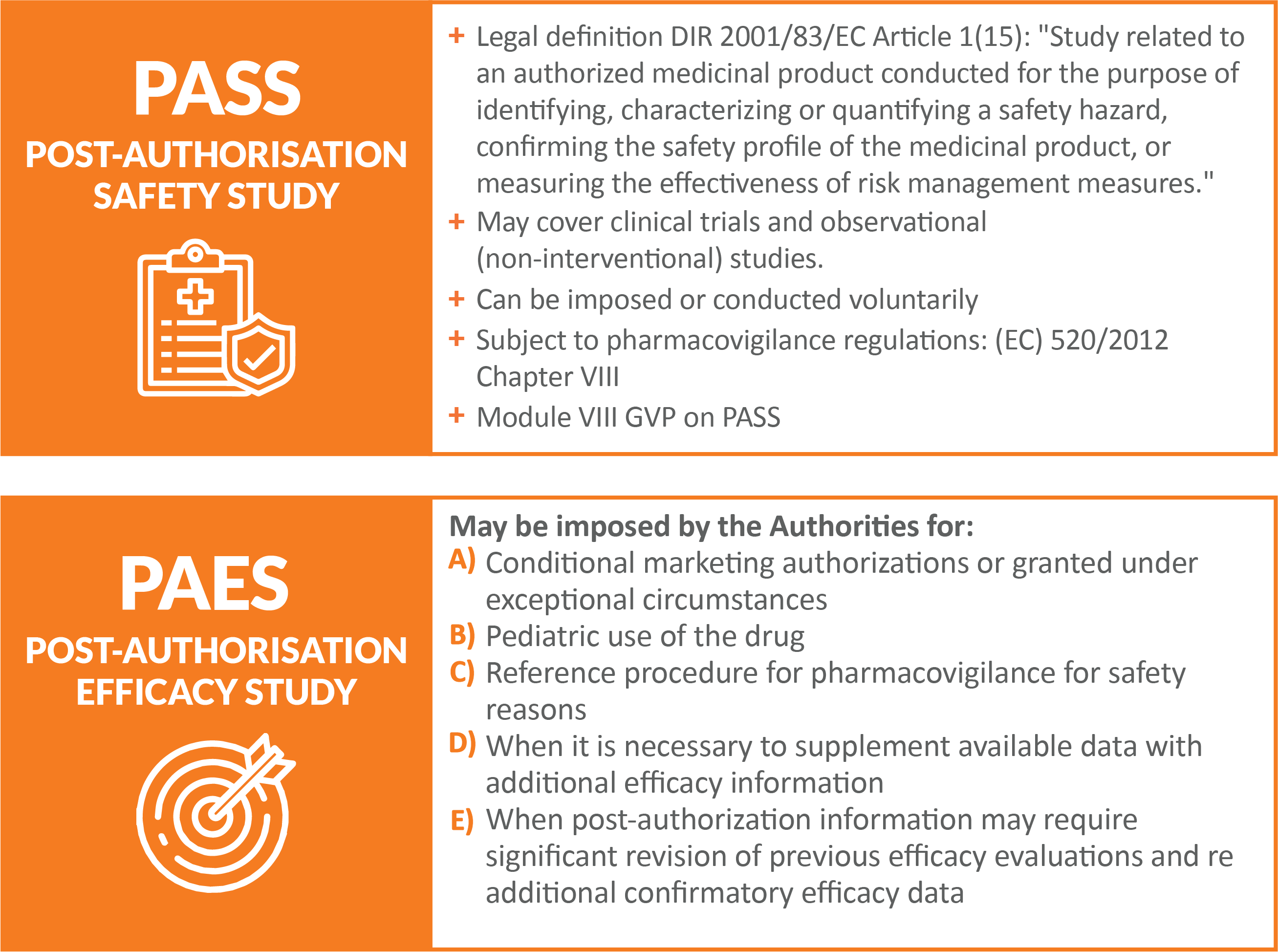
- Monitoring the safety of medicinal products requires a major commitment from manufacturers and authorities. PASS and PEAS studies are often necessary to validate the risk/benefit of new products.
- PASS studies of Pfizer-BioNTech and Moderna vaccines show significant gaps in available information. In addition, many of the studies are not traceable from the documents that should be present of the EMA portal.
- The characteristic of these studies makes clear the need for rigor and compliance with “best practices” in their conduct, with the involvement of specialized organizations to collect, manage and analyze data with high levels of quality and transparency.
- Thanks to digitization and accessibility in authority portals, these studies are now under greater scrutiny.
- It is suggested that manufacturers plan studies carefully so as to achieve the objectives of scientific demonstration and consumer assurance.
THE POST-AUTHORIZATION STUDIES
Medicinal products are monitored after they are marketed to confirm, characterize, and quantify their safety parameters. In Italy, the Italian Medicines Agency (Aifa) among its activities, oversees the safe use of medicines to ensure that the benefit-risk ratio of each drug is favorable.
For new drugs, some adverse reactions emerge only when these products are introduced into daily clinical practice and used on a real, uncontrolled population. Thus, clinical trials are often necessary to be able to identify any additional risks or assess uncertainties about benefits. These post-authorization studies focused on safety (PASS) or efficacy (PAES), are particularly critical in cases where the drug has gained rapid approval through emergency procedures, as in the case of Covid-19 vaccines.
MANY UNKNOWNS ABOUT RISK/BENEFIT
Covid-19 vaccines are the most recent and important example of accelerated licensing. Vaccines from Pfizer-BioNTech, Moderna, AstraZeneca and Janssen in Europe have received “conditional” approvals, based on less evidence than traditionally required for full approval on their safety and efficacy.
In fact, the randomized trials conducted to obtain approval were evaluated after a short follow-up period of a few months and on a relatively small number of patients. In December 2020, the US Fda authority also listed important uncertainties: whether the vaccine reduced the risk of hospitalization, ICU admission, mortality, or whether it provided protection in patients at high risk of severe Covid-19.
In fact, at the time of the launch of mass-use vaccines, very limited data were available on the risk of uncommon adverse events, as well as potential medium- or long-term harms, leaving some of the most important questions about efficacy and safety unanswered at the post-authorization stage.
PASS STUDIES FOR ANTI-COVID-19 VACCINES
The “conditional” authorization obtained for vaccines ensures that all of the manufacturer’s post-marketing obligations are legally binding and assessed by Ema. The requirements are codified in risk management plans developed by the manufacturer and agreed with the authority prior to authorization. These authority-mandated post-authorization studies contribute to the EU pharmacovigilance system in a complementary way to spontaneous adverse event reporting through pharmacovigilance platforms: EudraVigilance in Europe and Yellow Card Scheme in the UK. In the U.S., the Vaers database for vaccines was created specifically for the Covid-19 emergency; not only health care providers but also patients can enter adverse event reports.
Vaccine manufacturers Pfizer-BioNTech and Moderna have planned to conduct 13 and 8 post-authorization studies, respectively, to evaluate important unknowns including: risk of vaccine-associated disease; to examine effects in pregnant and lactating women, immunocompromised, frail people, or those with comorbidities or autoimmune or inflammatory diseases; to observe potential interaction between different vaccines; and to provide long-term safety data.
However, a recent article, published in the British Medical Journal, evaluated 21 PASS studies of Pfizer-BioNTech and Moderna vaccines. The authors found significant gaps in the available information, for example, the protocol was not available for more than half of the studies; four studies were dedicated to new vaccine versions or to studying booster effects in healthy and immunocompromised patients. Many of the studies were not traceable from the documents that are supposed to be in the Ema portal: this indicates that they were not conducted or remained in the early stages.
The case of Covid-19 vaccines highlights that manufacturers prioritize PASS studies not to assess safety, but to develop new vaccines or gain approval for additional doses of current vaccines. The need for data on major complications such as hospital and ICU admissions or death in moderate or high risk populations is overlooked.
Safety data therefore emerge overwhelmingly from the pharmacovigilance system. In Italy, AIFA published the “Annual Report on the Safety of Anti-Covid-19 Vaccines” containing an analysis of 134,361 reports of suspected adverse reactions following vaccination sent to the National Pharmacovigilance Network between December 27, 2020 and March 26, 2022. To estimate vaccine adverse reactions, which are based on voluntary reports and without uniformity of information, it must be taken into account that data provided by national or supranational government agencies have less homogeneity and reliability than a well-executed PASS study.
ACCELERATED APPROVALS AND HEALTH RISK
The landscape for conducting post-authorization studies of vaccines is very complex and requires considerable effort on the part of the marketing authorization holder.
To support the development of essential medicines, Ema has devised accelerated access pathways through the PRIority MEdicine (Prime) scheme[1]. The Prime scheme was launched in March 2016, improving early interaction and dialogue with regulatory authorities and thereby accelerating the regulatory assessment process for marketing authorization (MA)[2].
Although such regulatory tools can help meet therapeutic needs more quickly, they lead to greater uncertainty about the risk-benefit ratio of the product at the time of marketing and require robust strategies to support the overall safety profile. Ema therefore requires AIC holders to conduct extensive post-marketing studies (PASS) in order to generate more robust evidence.
EUROPEAN REGISTER OF POST-AUTHORIZATION STUDIES
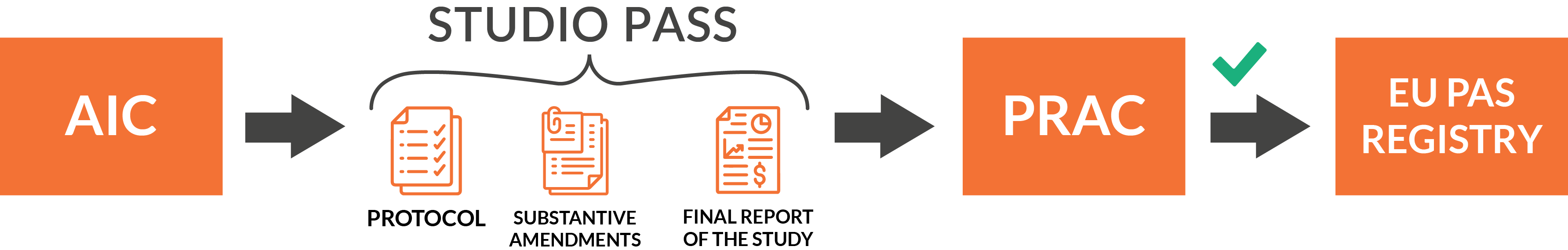
To date, Ema is the only regulatory agency that provides access to data and documents from mandatory post-authorization studies, and it makes summaries of study protocols and reports available through the EU Post-Authorization Studies Electronic Registry (EU PAS Registry). This is a publicly available repository supported by Ema through the European Network of Pharmacoepidemiology and Pharmacovigilance Centers (ENCePP).

Indeed, current pharmacovigilance legislation in Europe requires that PASS study protocols and the summary of results of such studies imposed as a condition of marketing authorization (category 1) or as a specific obligation in the context of a marketing authorization in exceptional circumstances (category 2) be made public. This registry is also intended to host studies that are not imposed but generated voluntarily as part of the Risk Management Plan (category 3) and all observational studies performed on authorized medicines, including efficacy studies (PAES). The EU PAS registry is the platform through which all these studies are accessible online.
The goal of the EU PAS registry is to improve transparency on studies, ensure the appropriateness of published data, promote information exchange and collaboration among stakeholders, including academia, sponsors, and regulatory bodies, and ensure compliance with the requirements of EU pharmacovigilance legislation.
An article published by ENCePP WG3 analyzed 1426 studies registered in the EU PAS registry from the beginning of its implementation until 2018. The analysis shows that among the 1380 studies entered with adequate information, 65.4 percent were requested by a regulatory body, 82 percent were funded by a pharmaceutical company, and 58.1 percent published the protocol. 86% of the studies had an observational design, 2.1% were interventional clinical trials, and 26% of the 1227 observational studies were multi-database studies. The characteristic of these studies makes it clear that there is a need for rigor and compliance with “best practices” in their conduct, with the intervention of the organizations in charge of data collection, management and analysis with high levels of quality and transparency.
TRANSPARENCY IS FUNDAMENTAL
To conduct a PASS study, the AIC holder must submit the protocol, any substantial amendments to the protocol prior to their implementation, and the final study report to the Pharmacovigilance Risk Assessment Committee (Prac).
The high level of scrutiny applied by regulatory authorities to protocols and results is justified by the potential conflicts of interest that may exist when pharmaceutical companies are asked to conduct studies whose results could adversely affect their products. For this reason, Ema strongly recommends in Gvp that companies conducting a PASS adhere to the “Code of Conduct” published by ENCePP. The “Code of Conduct” aims to promote scientific independence and transparency in the relationship between researchers and funders throughout the research process and, as a result, to strengthen public and scientific community trust in the integrity and value of research. The “Code of Conduct” addresses many issues including the rights and obligations of researchers and the funder, declarations of interest, the research contract, ownership and reporting of results, and publications.
THE FUTURE IN EUROPE
Ema launched its new Ctis information system in January 2022 as a single access point for clinical trial data submission in the EU. Information in this system will be made public, subject to EU transparency rules. The system is already fully functional and researchers can participate in the training program on how to use it. Ema is also implementing data analysis and a network of “real world” studies to generate comprehensive evidence on drug safety and efficacy from health databases. Interestingly, the same phenomenon is taking place in the medical technology sector, for which the Eudamed database for medical devices and in vitro diagnostics will for the first time enable centralization of all information in a centralized manner, including safety information.
The increasing digitization of health care systems in Europe and the availability of new information technology platforms dedicated to optimizing efficiency and quality in the conduct of clinical trials presents an important opportunity for innovative drug manufacturers to carefully plan post-marketing studies in order to generate stronger evidence on the safety and efficacy of their products, building consumer confidence. The characteristic of these studies makes clear the need for rigor and compliance with “best practices” in their conduct, with the involvement of specialized organizations to collect, manage and analyze data with high levels of quality and transparency.
[1] Neez E, Hwang T, Sahoo SA, Naci H, European Medicines Agency’s Priority Medicines (PRIME) scheme at 2 years: An evaluation of clinical studies supporting eligible drugs, Clin. Pharmacol. Ther. 2019; 1669
[2] https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/enhanced-early-dialogue-facilitate-accelerated-assessment-priority-medicines-prime_en.pdf







Leave A Comment